Copyright : Arkowitz, Avella, Braendle, Delaunay, Descombes, Fonta, Géminard, Plouraboué, Slagmulder, Van de Bor.
Copyright : Arkowitz, Avella, Braendle, Delaunay, Descombes, Fonta, Géminard, Plouraboué, Slagmulder, Van de Bor.Please note that all Team Presentation Forms below have been updated in the 2015 Pamphlet : here
-
Abad Pierre – Plant nematode interactions
ABAD Pierre

ABAD Pierre, INRA Group Leader ISA pierre.abad@sophia.inra.fr 0492386602
Website
Plant nematode interactions
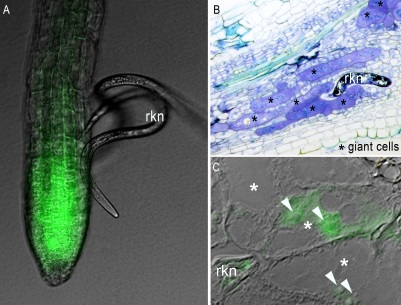 Root-knot nematodes (rkn) induce host giant cells. A. Arabidopsis GFP line & rkn larvae; B. Giant cells; C. Rkn proteins target host nuclei
Root-knot nematodes (rkn) induce host giant cells. A. Arabidopsis GFP line & rkn larvae; B. Giant cells; C. Rkn proteins target host nucleiPlant-parasitic nematodes have evolved sophisticated strategies for exploiting plants. These pathogens establish a long-lasting, intimate relationship with their hosts. Interaction involves the redifferentiation of root cells into specialised multinucleate feeding cells, named giant cells that provide nutrients to the parasite. Plant parasitic nematodes inject proteins into the host to modulate both plant developmental and immune signalling pathways. Our main model system is the root-knot nematode (RKN) Meloidogyne incognita. RKN is one of the most devastating plant pathogens able to infect thousand of plant species. We coordinated the international network on the genome sequence. We develop an integrated approach on both partners of the interaction combining plant pathology, plant biotechnology, molecular genetics, comparative genomics and advanced microscopy. Our aim is to identify key players involved in signalling pathways of host–parasite molecular dialogue i.e. the targets of RKN effectors in roots of the plant model Arabidopsis, the molecular mechanisms underlying the trade-off between development and immunity in plant roots and nematode adaptation to the host. Research into nematode parasitism thus tackles fundamental questions in plant development and plant pathology with the aims to design sustainable and environmentally friendly new methods to control nematodes. Selected Publications
1. A root-knot nematode-secreted protein is injected into giant cells and targeted to the nuclei Jaouannet M, Perfus-Barbeoch L, Deleury E, Magliano M, Engler G, Vieira P, Danchin EG, Da Rocha M, Coquillard P, Abad P, and Rosso MN (2012) New Phytol 194, 924-931 2. Multiple lateral gene transfers and duplications have promoted plant parasitism ability in nematodes Danchin EGJ, Rosso M-N, Vieira P, de Almeida-Engler J, Coutinho P, Henrissat B, Abad P (2010) Proc. Natl. Acad. Sci. USA. 107, 17651-17656 3. MAP65-3 microtubule-associated protein is essential for nematode-induced giant cell ontogenesis in Arabidopsis Caillaud MC, Lecomte P, Jammes F, Quentin M, Pagnotta S, Andrio E, de Almeida Engler JD, Marfaing N, Gounon, P Abad P, and Favery B (2008) Plant Cell 20, 423-437 4. Genome sequence of the metazoan plant-parasitic nematode Meloidogyne incognita Abad P, Gouzy J, Aury J-M, Castagnone-Sereno P, Danchin EGJ, Deleury E, Perfus-Barbeoch L et al (2008) Nature Biotech 26, 909-915 5. Arabidopsis formin AtFH6 is a plasma membrane–associated protein upregulated in giant cells induced by parasitic nematodes Favery B, Chelysheva L, Lebris M, Jammes F, De Almeida-Engler J, Marmagne A, Lecomte P Vaury C, Arkowitz RA, Abad P (2004) Plant Cell 16, 2529-2540 Awards 2012: Prize of the Academy of Sciences – Grand prix de l’Académie des Sciences Roger-Jean et Chantal Gautheret – Plant Physiology & Biotechnology
-
Antonny Bruno – Dynamics of lipid membranes and protein coats
ANTONNY Bruno

ANTONNY Bruno, CNRS Group Leader IPMC antonny@ipmc.cnrs.fr 04 93 95 77 75 http://www.ipmc.cnrs.fr/?page=antonny 
Dynamics of lipid membranes and protein coats
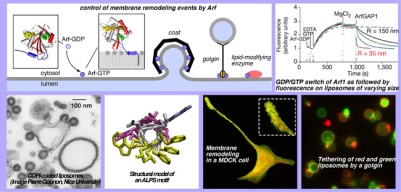 Studying membrane remodeling reactions using biochemical, biophysical, computer-based and cellular approaches
Studying membrane remodeling reactions using biochemical, biophysical, computer-based and cellular approachesVarious proteins remodel the membranes of organelles involved in intracellular transport. Protein coats deform membranes to promote the budding of vesicles. Golgins, sort of molecular strings, tether vesicles to restrict their diffusion. Lipid transporters adjust the membrane composition. Although very different, most of these mechanisms are controlled by small G proteins of the Arf family and by the physical chemistry of membranes.We study these mechanisms through molecular, cellular and in silico approaches. With original assays based on fluorescence and light scattering, we follow elementary reactions such as the assembly cycle of protein coats, the tethering of liposomes by a golgin or the transfer of lipids. With fluorescence light microscopy and electron microscopy, we visualize these events in cells and in reconstituted systems. With molecular dynamics, we describe at the atomic level how specific protein motifs sense the chemistry and curvature of lipid membranes.Recent achievements: – Discovery of a general motif to sense membrane curvature: the ALPS motif – Elucidation of the mechanism by which alpha synuclein, a protein involved in Parkinson disease, recognizes the curvature of endocytic vesicles. – Discovery of a mechanism by which cholesterol is transported through the coupled transfer and hydrolysis of phosphoinositides. Selected Publications
1. Amphipathic lipid packing sensor motifs: probing bilayer defects with hydrophobic residues Vanni S, Vamparys L, Gautier R, Drin G, Etchebest C, Fuchs PF and Antonny B (2013) Biophys J 104, 575-584 2. Curvature, lipid packing, and electrostatics of membrane organelles: defining cellular territories in determining specificity Bigay J and Antonny B (2013) Dev Cell 23, 886-895 3. Osh4p exchanges sterols for phosphatidylinositol 4-phosphate between lipid bilayers de Saint-Jean M, Delfosse V, Douguet D, Chicanne G, Payrastre B, Bourguet W, Antonny B and Drin G (2011) J Cell Biol 195, 965-978 4. alpha-Synuclein and ALPS motifs are membrane curvature sensors whose contrasting chemistry mediates selective vesicle binding Pranke IM, Morello V, Bigay J, Gibson K, Verbavatz JM, Antonny B and Jackson CL (2011) J Cell Biol 194, 89-103 5. Asymmetric tethering of flat and curved lipid membranes by a golgin Drin G, Morello V, Casella JF, Gounon P and Antonny B (2008) Science 320, 670-673 Awards 2010: CNRS bronze medal, Guillaume Drin 2010: ERC advanced grant, Bruno Antonny 2009: CNRS silver medal, Bruno Antonny 2008: EMBO member, Bruno Antonny
-
Arkowitz Robert – Polarized growth in yeast
ARKOWITZ Robert

ARKOWITZ Robert, CNRS Group Leader iBV arkowitz@unice.fr 0492076425 http://ibv.unice.fr/EN/equipe/arkowitz.php 
Polarized growth in yeast
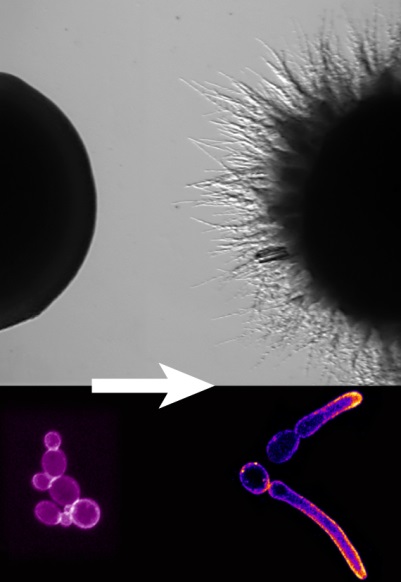 The yeast (left) to filamentous (right) transition is critical for Candida albicans virulence. Colonies (top) & individual cells (bottom).
The yeast (left) to filamentous (right) transition is critical for Candida albicans virulence. Colonies (top) & individual cells (bottom).Polarized growth is essential for both internal organization of cells and generation of complex multi-cellular structures. Our main interest is how cells spatially and temporally regulate their growth.Our goal is to understand the mechanisms of polarized growth and cell morphogenesis in yeast. The yeast Saccharomyces cerevisiae reproduces during its haploid mitotic life cycle by budding. When haploid cells of the opposite mating type come in contact they direct their growth towards one another, forming pear shaped polarized cells, which ultimately fuse to form diploids. Upon nitrogen starvation diploid yeast switch from unicellular budding to a filamentous form comprised of chains of elongated cells. The opportunistic human pathogenic yeast Candida albicans switches from an oval form to an invasive, filamentous hyphal form, a process critical for its pathogenicity.In these 3 growth processes asymmetric cell growth is accomplished by polarization of the actin cytoskeleton and subsequent localized growth by directed membrane traffic. During budding, polarized growth is initiated by internal signals whereas during mating and hyphal formation, it is dictated by external signals. We are interested in how internal and external signals are linked to site-specific growth, with particular focus on the roles of small G-proteins and phospholipids both in S. cerevisiae and C. albicans. Selected Publications
1. Phosphoinositides-bis-phosphate is required for Saccharomyces cerevisiae invasive growth.Guillas I, Vernay A, Vitagliano JJ and Arkowitz RA. (2013) J Cell Sci in press 2. A steep phosphoinositide bis-phosphate gradient forms during fungal filamentous growth Vernay A, Schaub S, Guillas I, Bassilana M and Arkowitz, RA (2012) J Cell Biol 198, 711-730 3. Polarized growth in fungi: symmetry breaking and hyphal formation Arkowitz RA and Bassilana M (2011) Semin Cell Dev Biol 22, 806-815 4. The Candida albicans ELMO homologue functions together with Rac1 and Dck1, upstream of the MAP Kinase Cek1, in invasive filamentous growth Hope H, Schmauch C, Arkowitz RA and Bassilana M (2010) Mol Microbiol 76, 1572-1590 5. Activation of Rac1 by the Guanine Nucleotide Exchange Factor Dck1 Is Required for Invasive Filamentous Growth in the Pathogen Candida albicans Hope H, Bogliolo S, Arkowitz RA and Bassilana M (2008) Mol Biol Cell 19, 3638-3651 Awards 2013: Marie Curie ITN Consortium 2001-2010: Fondation pour la Recherche Médicale – BNP Paribas Award 2002-2004: La Ligue Contre le Cancer – Selected Team 2001-: EMBO Young Investigator Programme 2000-2003: CNRS ATIP (start up package)
-
Auberger Patrick – Cell Death, Differentiation, Inflammation and Cancer
AUBERGER Patrick

AUBERGER Patrick, Inserm Group Leader C3M auberger@unice.fr 04 89 06 43 11 http://www.unice.fr/c3m/EN/Equipe2.html 
Cell deaths, Differentiation, Inflammation and Cancer
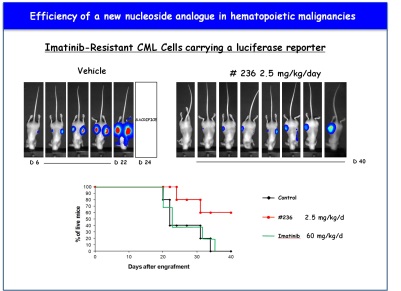 Effect of a new triazole nucleoside (#236) on tumor formation in athymic mice. Overall survival rates in treated mice .
Effect of a new triazole nucleoside (#236) on tumor formation in athymic mice. Overall survival rates in treated mice .Our team investigates the deregulation of cell death processes and autophagy in hematopoietic malignancies and during skin inflammatory processes. We are also developping new alternative therapeutic strategies to circumvent the resistance to conventionnal chemotherapies focusing our interest on Leukemia, Myelodysplastic Syndromes and Myeloma. We are also interested in the role of teh tyrosine kinase Lyn and of inflammatory caspases in the physiopathology of human psoriasis. In this context our mains focus of interest are: 1- The mechanisms of resistance to tyrosine kinase inhibitors in Chronic Myelogenous Leukemia (CML) 2- The mechanisms of resistance to nucleoside analogues in MyeloDysplastic Syndromes (MDS) and Acute Myeloid Leukemia (AML) 3- The role of BCL2L10 (BCL-B), an anti-apoptotic member of the BCL2 family in the homeostasis of plasma cells and the physiopathology of Multiple Myeloma (MM) 4- Role of the inflammatory caspases in the context of skin disorders such as prosiasis: pre-clinical and clinical study. 5- Regulation of the pro-apoptotic function of a bcl-2 family member by oncogenic tyrosine kinases of the Src family. Our research project is at the Biology / Chemistry / Clinical interface and aims at validating new biomarkers and drug candidates for the treatment of some hematopoietic malignancies (CML, MDS, AML, MM) and psoriasis. Keywords : Autophagy, Cell Selected Publications
1. The caspase 6 derived N-terminal fragment of DJ-1 promotes apoptosis via increased ROS production Robert G, Puissant A, Dufies M, Marchetti S, Jacquel A, Cluzeau T, Colosetti P, Belhacene N, Kahle P, Da Costa CA, Luciano, F Checler, F and Auberger P (2012) Cell Death Differ 19, 1769-1778 2. Imatinib triggers mesenchymal-like conversion of CML cells associated with increased aggressiveness Puissant A, Dufies M, Fenouille N, Ben Sahra I, Jcquel A, Robert G, Cluzeau T, Deckert M, Tichet M, Cheli Y Cassuto JP, Raynaud S, Legros L, Pasquet JM, Mahon FX, Luciano F and Auberger P (2012) J Mol Cell Biol 4, 207-220 3. Autophagy is required for CSF-1induced macrophagic differentiation and acquisition of pahgocytic function Jacquel A, Obba S, Boyer L, Dufies M, Robert G, Gounon P, Lemichez E, Luciano F, Solary E and Auberger P (2012) Blood 119, 4527-4531 4. Cathepsin B release after imatinib-mediated lysosomal membrane permeabilization triggers BCR-ABL cleavage and elimination of chronic myelogenous leukemia cells Puissant A, colosetti P, Robert G, cassuto JP, Raynaud S and Auberger P (2010) Leukemia 24, 115-124 5. The caspase-cleaved form of LYN mediates a psoriatic-like inflammatory syndrome in mice Marchetti S, Gamas P, Belhacene N, Grosso S, Pradelli L, Colosetti P, Johansen C, Iversen L, Dekert M, Luciano P, Hofman P, Ortonne N, Khemis A Mari B, Ortonne JP, Ricci JE and Auberger P (2009) EMBO J 28, 2449-2460
-
Ballotti Robert – Biology and pathology of melanocytic cells: from cutaneous pigmentation to melanomas
BALLOTTI Robert

BALLOTTI Robert, Inserm Group Leader C3M ballotti@unice.fr 0489064332 http://www.unice.fr/c3m/EN/Equipe1.html 
Biology and pathology of melanocytic cells: from cutaneous pigmentation to melanomas
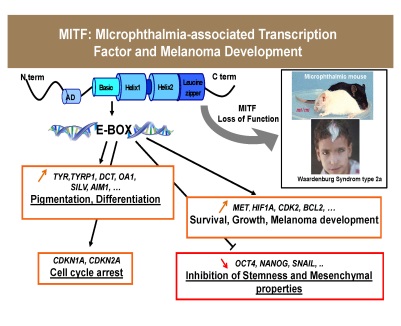 Schematic representation of MITF functions in melanocytes and melanomas
Schematic representation of MITF functions in melanocytes and melanomasOur projects are based on very recent and original data obtained in our laboratory, i.e., (i) the identification of a MITF-negative population endowed with melanoma-initiating properties; and (ii) the discovery of a new MITF mutant (E318K) that favours melanoma development.1) We will study the effects of stresses relevant to melanoma development on MITF SUMOylation. We will focus our attention on UV radiation and hypoxia, and then to study the role of MITF SUMOylation, we will compare, in melanoma cells or in mouse model expressing WT or E318K MITF, the effects (migration, invasion, tumorigenicity) of various stimuli identified in the first part of the project to modulate MITF SUMOylation.2) We identified a MITF-negative population, expressing stem cell markers (OCT4, NANOG), displaying high tumorigenic potential and slow growing rate that fits perfectly with the definition of tumor initiating cells. Taking advantage of a new cell surface marker for melanoma initiating cells that we identified recently, we will (i) purify the MIC population (ii) study their biological properties such as growth, migration, differentiation, tumorigenicity, drug resistance and response to environmental stimuli, (iii) Identify the repertoire of gene expressed in the melanoma initiating cells and (iv) validate this marker in human melanoma samples, in correlation the clinical outcome. Selected Publications
1. Major clinical response to a BRAF inhibitor in a patient with a BRAF L597R mutated melanoma Bahadoran, P., Allegra, M., Le Duff, F., Long-Mira, E., Hofman, P., Giacchero, D., Passeron, P., Lacour, JP., Ballotti, R. (2013) J Clin Oncol in press, 1-2 2. A SUMOylation-defective MITF germline mutation predisposes to melanoma and renal carcinoma Bertolotto, C., Lesueur, F., Giuliano, S., Strub, T., de Lichy, M., Bille, K., Dessen, P., d’Hayer, B., Mohamdi, H., Remenieras, A., Maubec, E., de la Fouchardiere, A., Molinie, V., Vabres, P., Dalle, S., Poulalhon, N., Martin-Denavit, T., Thomas, L., Andry-Benzaquen, P., Dupin, N., Boitier, F., Rossi, A., Perrot, J.L., Labeille, B., Robert, C., Escudier, B., Caron, O., Brugieres, L., Saule, S., Gardie, B., Gad, S., Richard, S., Couturier, J., Teh, B.T., Ghiorzo, P., Pastorino, L., Puig, S., Badenas, C., Olsson, H., Ingvar, C., Rouleau, E., Lidereau, R., Bahadoran, P., Vielh, P., Corda, E., Blanche, H., Zelenika, D., Galan, P., Aubin, F., Bachollet, B., Becuwe, C., Berthet, P., Bignon, Y.J., Bonadona, V., Bonafe, J.L., Bonnet-Dupeyron, M.N., Cambazard, F., Chevrant-Breton, J., Coupier, I., Dalac, S., Demange, L., d’Incan, M., Dugast, C., Faivre, L., Vincent-Fetita, L., Gauthier-Villars, M., Gilbert, B., Grange, F., Grob, J.J., Humbert, P., Janin, N., Joly, P., Kerob, D., Lasset, C., Leroux, D., Levang, J., Limacher, J.M., Livideanu, C., Longy, M., Lortholary, A., Stoppa-Lyonnet, D., Mansard, S., Mansuy, L., Marrou, K., Mateus, C., Maugard, C., Meyer, N., Nogues, C., Souteyrand, P., Venat-Bouvet, L., Zattara, H., Chaudru, V., Lenoir, G.M., Lathrop, M., Davidson, I., Avril, M.F., Demenais, F., Ballotti, R*., and Bressac-de Paillerets*, B (2011) Nature 480, 94-98 3. Hypoxia and MITF control metastatic behaviour in mouse and human melanoma cells Cheli, Y., Giuliano, S., Fenouille, N., Allegra, M., Hofman, V., Hofman, P., Bahadoran, P., Lacour, J.P., Tartare-Deckert, S., Bertolotto, C., and Ballotti, R (2012) Oncogene 31, 2461-2470 4. Senescent cells develop a PARP-1 and nuclear factor-{kappa}B-associated secretome (PNAS Ohanna, M., Giuliano, S., Bonet, C., Imbert, V., Hofman, V., Zangari, J., Bille, K., Robert, C., Bressac-de Paillerets, B., Hofman, P., Rocchi, S., Peyron, J.F., Lacour, J.P., Ballotti, R., and Bertolotto, C (2011) Genes & Dev 25, 1245-1261 5. Mitf is the key molecular switch between mouse or human melanoma initiating cells and their differentiated progeny Cheli, Y., Giuliano, S., Botton, T., Rocchi, S., Hofman, V., Hofman, P., Bahadoran, P., Bertolotto, C., and Ballotti, R (2011) Oncogene 30, 2307-2318
-
Barbry Pascal – Physiological Genomics of the Eukaryotes
BARBRY Pascal

BARBRY Pascal, CNRS Group Leader IPMC direction@ipmc.cnrs.fr0493957793 https://www.ipmc.cnrs.fr/cgi-bin/standard.cgi?descriptif=barbry.txt&dossier1=equipes&dossier2=barbry&site=inter&menu=1&ssmenu=2&lang=uk 
Physiological Genomics of the Eukaryotes
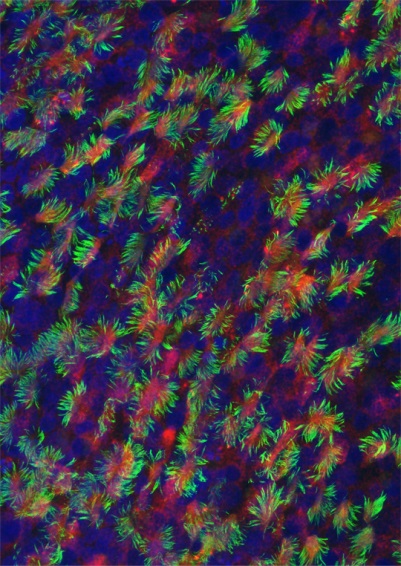 Immunolabelling of multiciliated airway epithelial cells (RED=basal bodies; GREEN=motille cilia; BLUE=nuclei)
Immunolabelling of multiciliated airway epithelial cells (RED=basal bodies; GREEN=motille cilia; BLUE=nuclei)Respiratory cells develop specific differentiation programs, for instance to synthesize hundreds of motile cilia at the apical surface of the airway epithelial cells. This allows an efficient protection of the respiratory tract mucosa against various external aggressions, and provides specific responses against stresses (allergens, bacteria, viruses, chemicals…). Airway epithelial cells represent a unique cellular model, which is central to important health questions (asthma, lung cancer, cystic fibrosis, fibroproliferative diseases,…). Our group is more specifically interested by the regulations by small regulatory RNAs that can take place in lung cells. In that context, we have recently reported the identification of microRNAs of the miR-34/miR-449 families as evolutionary conserved key regulators of vertebrate multiciliogenesis. We also found that miR-199a-5p behaves as a major regulator of tissue fibrosis with interesting therapeutic potency to treat fibroproliferative diseases. Our interest for dysregulated microRNAs expression during lung cancer and fibrosis led to investigate the impact of other microRNAs, such as miR-210 and miR-483, on cell metabolism, viability, apoptosis, migration and wound healing. For all these studies, our group develops new approaches in functional genomics and bioinformatics, with a special interest for high throughput sequencing. Selected Publications
1. CDC25A targeting by miR-483-3p decreases CCND-CDK4/6 assembly and contributes to cell cycle arrest. Bertero T, Gastaldi C, Bourget-Ponzio I, Mari B, Meneguzzi G, Barbry P, Ponzio G, Rezzonico R. (2013) Cell Death Diff 4, e544 2. miR-199a-5p is upregulated during fibrogenic response to tissue injury and mediates TGFbeta-induced lung fibroblast activation by targeting caveolin-1 Cardenas CLL, Henaoui IS, Courcot E, Roderburg C, Cauffiez C, Aubert S, Copin MC, Wallaert B, Glowacki F, Dewaeles E, Milosevic J, Maurizio J, Tedrow J, Marcet B, Lo-guidice JM, Kaminski N, Barbry P, Luedde T, Perrais M, Mari B, Pottier N. (2013) PLoS Genet 9(2), e1003291 3. Gene expression profiling reveals distinct epithelial phenotypes in child respiratory allergy Giovannini-Chami L, Marcet B, Moreilhon C, Chevalier B, Illie MI, LeBrigand K, Robbe-Sermesant K, Bourrier T, Michiels JF, Mari B, Crénesse D, Hofman P, de Blic J, Castillo L, Albertini M, Barbry P. (2012) Eur Resp J 39(5), 1197-205 4. Small RNA sequencing reveals miR-642a-3p as a novel adipocyte-specific microRNA and miR-30 as a key regulator of human adipogenesis. Zaragosi LE, Wdziekonski B, Le Brigand K, Villageois P, Mari B, Waldmann R, Dani C, Barbry P. (2011) Genome Biol 12(7) , R64 5. miR-449 microRNAs trigger vertebrate multiciliogenesis through direct repression of the Notch ligand Delta-like 1. Marcet B, Coraux C, Chevalier B, Luxardi G, Zaragosi LE, Robbe-Sermesant K, Jolly T, Cardinaud B, Moreilhon C, Giovannini-Chami L, Birembaut P, Waldmann R, Kodjabachian L, Barbry P. (2011) Nature Cell Biology. 13(6), 694-701 Awards 2011: CNRS PES award 2010: Coordinator of InDiGen, a component of France-Génomique
-
Bardoni Barbara – Physiopathology of intellectual disability
BARDONI Barbara

BARDONI Barbara, Inserm Group Leader IPMC bardoni@ipmc.cnrs.fr 0493957766/62/78 https://www.ipmc.cnrs.fr/cgi-bin/standard.cgi?descriptif=bardon 
Physiopathology of Intellectual disability
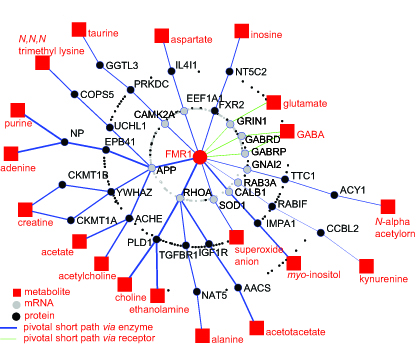 FMR1 integrated Metabolome and Interactome Mapping (iMIM) Network (Davidovic et al., Genom. Res. , 2013)
FMR1 integrated Metabolome and Interactome Mapping (iMIM) Network (Davidovic et al., Genom. Res. , 2013)Fragile X syndrome (FXS) is the most common form of inherited intellectual disability (ID) due to the silencing of the FMR1 gene, which encodes for FMRP, an RNA-binding protein involved in translational regulation at the synapse. In the past, we focused our studies on the characterization of partners (proteins and RNAs) of FMRP. More recently we addressed our efforts to the identification and characterisation of pathways involving FMRP function in adult neurons and during neuronal differentiation using chemical, bioinformatic, molecular biology, cellular and genomic analyses. Indeed, we identified novel pathways involving FMRP and that are perturbed in FXS. In some cases, modulation of these cellular signaling pathways improves the phenotype of the FXS mouse model. Our interests include the molecular mechanisms of action of FMRP and of its two paralogs FXR1P and FXR2P and search of biomarkers for FXS. We are also interested in the functional characterization of another gene silenced in another form of ID and autism, AFF/FMR2, coding for a transcription factor involved in Fos and Jun expression. We have defined its role also in splicing and we are currently searching for its RNA targets. We are studying the role of some miRNAs (targeting the mRNA of FMR1) in neuronal maturation by characterizing their mRNA targets and evaluating their implication in other forms of ID/autism. Selected Publications
1. A Novel Function of Fragile X Mental Retardation Protein in translational activation Bechara E, Didiot, MC, Melko M, Davidovic L, Bensaid M, Martin P, Castets M, Pognonec P, Khandjian E, Moine H, Bardoni B (2009) PLoS Biol.Jan 20 7(1), e16 2. Functional characterization of the AFF (AF4/FMR2) family of RNA binding proteins: insights into the molecular pathology of FRAXE intellectual disability. Melko M, Douguet D., Bensaid M, Zongaro S, Verheggen C, Gecz J, Bardoni B (2011) Hum. Mol. Genet. 20, 1873-1885 3. A metabolomic and system biology perspective on the brain of the Fragile X syndrome mouse model Davidovic L, Navratil V, Bonaccorso C.M., Catania M.V., Bardoni B, Dumas M (2011) Genome Res. 21, 2190-2202 4. The 3’ UTR of FMR1 mRNA is a target of miR-101, miR-129-5p and miR-221: implications for the molecular pathology of FXTAS at the synapse Zongaro S, Hukema R, D’Antoni S, Davidovic L, Barbry P, Catania M.V., Willemsen R, Mari B, Bardoni B (2013) Hum. Mol. Genet. 22, 1971-1982 5. A novel role for the RNA-binding protein FXR1P in myoblasts cell-cycle progression by modulating p21/Cdkn1a/Cip1/Waf1 mRNA stability. Davidovic L, Durand N, Khalfallah O, Tabet R, Barbry P, Mari B, Sacconi S, Moine H, Bardoni B (2013) Plos. Genet.Mar;9(3), e1003367 Awards 2004: CNRS ATIP 2007: CNRS ATIP PLUS
-
Besse Florence – Post-transcriptional control of axon growth and guidance in Drosophila
BESSE Florence

BESSE Florence, CNRS Group Leader iBV besse@unice.fr04 92 07 64 34 http://ibv.unice.fr/EN/equipe/besse.php 
Post-transcriptional control of axon growth and guidance in Drosophila
 3D reconstruction of a wild-type axonal tree
3D reconstruction of a wild-type axonal treeWhile post-transcriptional regulation of mRNAs has recently emerged as a major step in the spatial and temporal regulation of gene expression, very few studies have analyzed its underlying mechanisms and biological functions in vivo, during CNS maturation. Intracellular targeting of mRNAs coupled to local translation is a major component of the post-transcriptional toolkit, and is a powerful means to asymmetrically accumulate protein products within polarized cells. Ten years ago, local translation of transcripts localized to axons has been shown to be essential for growth cone turning in response to guidance cues in cultured neurons. Since then, several mRNAs have been shown to be recruited to growing axons and translated locally. Strikingly, the biological relevance of this process remains to be demonstrated in vivo, and the underlying molecular mechanisms are still largely unknown. We are using Drosophila CNS neurons as a genetically tractable system to 1) identify the components (mRNAs and associated proteins) of axonally-transported ribonucleoprotein complexes (RNP), and 2) characterize their function and regulation during development. To this end, we are combining different approaches including genetic screens, biochemical purifications, live imaging and bioinformatic analyses. Selected Publications
1. Principles and roles of mRNA localization in animal development. Medioni C., Mowry K. and Besse F. (2012) Development 139(18), 3263-76. 2. Drosophila PTB/hnRNPI promotes formation of high-order oskar RNP complexes and represses oskar translation. Besse F.*, Lopez-de-Quinto S.*, Marchand V., Trucco A. and Ephrussi A. (2009) Genes Dev. 23(2), 195-207 3. Translational control of localized mRNA: restricting protein synthesis in space and time. Besse F. and Ephrussi A. (2008) Nat Rev Mol Cell. Biol. 9(12), 971-80 4. The Ig-CAM Basigin controls compartmentalization and vesicle release at Drosophila synapses. Besse F., Mertel S., Kittel R., Wichman C., Rasse T, Sigrist S. and Ephrussi A. (2007) J. Cell. Biol. 177(5), 843-55. Awards 2009: HFSP Career Development Award 2008: ATIP CNRS
-
Braendle Christian – Gene-environment interactions in development and evolution
BRAENDLE Christian

BRAENDLE Christian, CNRS Group Leader iBV braendle@unice.fr 04 92 07 68 97 http://ibv.unice.fr/EN/equipe/braendle.php 
Gene-environement interactions in development and evolution
 The study system: the Caenorhabditis germline
The study system: the Caenorhabditis germlineHow does an organism adjust its reproduction and underlying developmental processes in response to environmental variation? How do such environmental responses evolve, given that populations and species have adapted to contrasted ecological niches? We address these questions by studying how molecular and cellular processes of the Caenorhabditis germline respond to environmental variation and how such responses evolve. The objective is to conduct an integrative analysis of such germline plasticity and its evolution by characterizing the processes of germ cell proliferation, gamete differentiation and maturation, which ultimately define the reproductive output of the organism. Specifically, we aim (1) to quantify plasticity and genotype-by-environment interactions for molecular and life history phenotypes in different Caenorhabditis species and isolates; (2) to determine the mechanisms underlying plastic phenotypic responses to environmental variation; (3) to identify molecular changes underlying genotype-by-environment interactions, using QTL mapping approaches and developmental genetics; (4) to track the evolution of plasticity in variable environments using experimental evolution; (5) to search for ecological correlates of the observed genotype-by-environment interactions by studying natural Caenorhabditis populations and defining their ecological context. Selected Publications
1. Species richness, distribution and genetic diversity of Caenorhabditis nematodes in a remote tropical rainforest Félix MA, Jovelin R, Ferrari C, Han S, Cho YR, Andersen EC, Cutter AD and Braendle C (2013) BMC Evol Biol 13, 10 2. Pheromones: Evolving language of chemical communication in nematodes Braendle C (2012) Curr Biol 22, R294-296 3. Bias and evolution of the mutationally accessible phenotypic space in a developmental system Braendle C, Baer C and Félix MA (2010) PLoS Genetics -, e1000877 4. Plasticity and errors of a robust developmental system in different environments Braendle C and Félix MA (2008) Dev Cell 15, 714-724 Awards 2010: Schlumberger Award 2008: ATIP
-
Braud Véronique/Anjuère Fabienne – Immune regulation at muco-cutaneous surfaces
BRAUD Véronique/ANJUERE Fabienne

BRAUD Véronique, CNRS Group Leader IPMC braud@ipmc.cnrs.fr 0493957771 http://www.ipmc.cnrs.fr 
ANJUERE Fabienne, Inserm Group Leader IPMC anjuere@ipmc.cnrs.fr 0493957771 http://www.ipmc.cnrs.fr 
Immune regulation at muco-cutaneous surfaces
 Appropriate immunity at epithelial sites contributes to host integrity
Appropriate immunity at epithelial sites contributes to host integrityNon-melanoma skin carcinomas are the most frequent human cancers worldwide. Malignant transformation of epithelial tissues is controlled by immune mechanisms in place within the epithelium itself. A fine-tune dialog between epithelial and immune cells is crucial to resolve local injury/damage caused to keratinocytes by external aggressions.Our research will therefore aim to provide a better understanding of the molecular and cellular responses of both the epithelium and the tissue-associated innate immune cells during the different stages of carcinoma development. We have obtained an extensive gene signature of innate immune cells and epithelial tumors at different stages of carcinoma development. Ongoing studies focus on the study of candidate genes and signalling pathways regulating carcinogenesis. The knowledge gained from these studies will allow the building of an integrative genomic picture of the tumor within its immune microenvironment and the identification of therapeutic targets not only for skin carcinomas such as basal-cell carcinomas (BCC) and more aggressive squamous cell carcinomas (SCC) but also for other epithelial cancers that are often refractory to treatments. Selected Publications
1. Antigen-bearing dendritic cells from the sublingual mucosa recirculate to distant systemic lymphoid organs to prime mucosal CD8 T. Hervouet C , Luci C, Bekri S, Juhel T, Bihl F, Braud VM, Czerkinsky and F Anjuère (2013) Mucosal Immunol in press, – 2. Induction of LLT1 cell surface expression by pathogens and IFN-g contributes to modulate immune responses. Germain C, Meier A, Jensen T, Knapnougel P, Poupon G, Lazzari A, Neisig A, Håkansson K, Dong T, Wagtmann N, Galsgaard ED, Spee P, and V. M. Braud (2011) J Biol Chem 286, 37964-37975 3. Langerhans cells prime IL-17-producing T cells and dampen genital cytotoxic responses following mucosal immunization Hervouet C, Luci C, Rol N, Rousseau D, Kissenpfennig A, Malissen B, Czerkinsky C, Anjuère F (2010) J Immunol 184(9) , 4842-4851 4. Dendritic cell-mediated induction of cytotoxic responses following intravaginal immunization with the non-toxic B subunit of cholera toxin. Luci C, Hervouet C, Rousseau D, Holmgren J, Czerkinsky C and Anjuère F. (2006) J Immunol 176, 2749-2757 5. Cutting Edge: Lectin-Like Transcript 1 Is a Ligand for the CD161 Receptor Aldemir H, Prod’homme V, Dumaurier MJ, Retiere C, Poupon G, Cazareth J, Bihl F, and Braud VM (2005) J Immunol 175, 7791-7795 Awards 2002: Price from the Fondation pour la Recherche Médicale: V. Braud 2000-2003: Young investigator fellowship ATIP BLANCHE CNRS: V. Braud
-
Chaboissier Marie-Christine – Genetics of sex determination and fertility
CHABOISSIER Marie-Christine

CHABOISSIER Marie-Christine, Inserm Group Leader iBV marie-christine.chaboissier@unice.fr0492076451 http://ibv.unice.fr/EN/equipe/chaboissier.php 
Genetics of sex determination and fertility
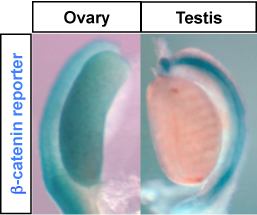 Rspo1/beta-catenin signaling and sex determination
Rspo1/beta-catenin signaling and sex determinationDisorders of sexual differentiation (DSD) are frequent diseases that present considerable challenges for physicians, parents and affected individuals. These challenges include surgical correction, management and, in some cases, gender assignment. Unfortunately a large fraction of human cases of DSD are unexplained, despite substantial progress in our understanding of the molecular regulation of testicular and ovarian differentiation and maintenance. Our group aims at identifying new factors involved in sex detrmination with the goal of developing new genetic tests for individuals with DSD. Using mouse models, we have contributed to show that sex determination depends on a fine tuned balance between the SRY/SOX9 pathway, which precipitates testis differentiation, and the R-spondin/beta-catenin signaling pathway involved in ovarian differentiation. Our aim is now to decipher how these pathways and newly identified ones regulate sexual differentiation in normal and pathological conditions. Selected Publications
1. Testicular differentiation occurs in absence of R-spondin1 and Sox9 in mouse sex reversals. Lavery R, Chassot AA, Pauper E, Gregoire EP, Klopfenstein M, de Rooij DG, Mark M, Schedl A, Ghyselinck NB. and Chaboissier MC (2012) PLoS Genet & Faculty 1000 e, 1003170 2. WNT4 and RSPO1 together are required for cell proliferation in the early mouse gonad. Chassot AA, Bradford ST, Auguste A, Gregoire EP, Pailhoux E, de Rooij DG, Schedl A and Chaboissier MC (2012) Development 139, 4461-4472 3. XY Sox9 loss-of-function mouse mutants show complete sex reversal and can produce fertile XY oocytes Lavery R, Lardenois A, Ranc-Jianmotamedi F, Pauper E, Gregoire EP, Vigier C, Moreilhon C, Primig M and Chaboissier MC (2011) Dev Biol & Faculty 1000 354, 111-122 4. Activation of b-catenin signalling by Rspo1 controls differentiation of mammalian ovary. Chassot AA, Ranc F, Gregoire E, Roepers-Gajadien HL, Camerino G, de Rooij DG, Schedl A and Chaboissier MCFunctional analysis of Sox8 and Sox9 during sex determination in the mouse. (2008) Human Mol Genet & Faculty 1000 17, 1264-1277 5. R-spondin1 plays an essential role in sex determination, skin differentiation and malignancy. Parma P, Radi O, Vidal VPI, Chaboissier MC, Dellambra E, Valentini S, Guerra L, Schedl A, and Camerino G. (2006) Nat Genet & Faculty 1000 38, 1304-1309 Awards 2008: Albert Sézary Prize for Medicine (to AA Chassot, post doc for R-spondin1
-
Collombat Patrick – Diabetes Genetics
COLLOMBAT Patrick

COLLOMBAT Patrick,Inserm Group Leader iBV collombat@unice.fr0492076416 http://collombat.com 
Diabetes Genetics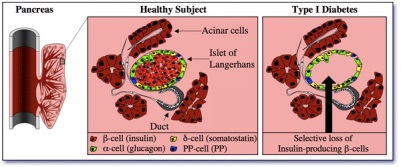 Type I diabetes in mouse and human
Type I diabetes in mouse and humanOur group is involved in understanding the molecular mechanisms underlying Diabetes and finding ways to counter this condition. Both Type I Diabetes (insulinodependent) and Type II (non insulinodependent) diabetes ultimately result in the selective loss of insulin-producing beta-cells in the endocrine pancreas (Figure). The subsequent lack in insulin hormone induces a blood hyperglycemia that may be attenuated by daily injection of exogenous insulin hormone. Nevertheless, despite the best of actual therapies, Type 1 Diabetic patients display a shortened life expectancy as compared to their healthy counterparts.We belong to a NIH-/JDRF-funded consortium whose goal is to regenerate lost beta-cells in the context of type 1 diabetes. Using the mouse as a model, we have identified two transcription factors, Arx and Pax4, playing a crucial role in the genesis of the different endocrine cell subtypes, including insulin-secreting beta-cells. Importantly, the forced expression of Pax4 in specific cells of the pancreas can induce their proliferation and conversion in beta-cells. Of note, these cells are functional and can counter several cycles of chemically-induced diabetes.Our group is now looking into ways to “drug » these beta-cell regeneration processes and determine whether our results could be applied to human. Selected Publications
1. Adult duct-lining cells can reprogram into β-like cells able to counter repeated cycles of toxin-induced diabetes Al-Hasani K, Pfeifer A, Courtney M, Ben-Othman N, Gjernes E, Vieira A, Druelle N, Avolio F, Ravassard P, Leuckx G, Lacas-Gervais S, Ambrosetti D, Benizri E, Hecksher-Sorensen J, Gounon P, Ferrer J, Gradwohl G, Heimberg H, Mansouri M, Collombat P (2013) Dev Cell In press, In press 2. The homeodomain-containing transcription factors arx and pax4 control enteroendocrine subtype specification in mice Beucher A, Gjernes E, Collin C, Courtney M, Meunier A, Collombat P, and Gradwohl G (2009) Plos One 7, Pe36449 3. The ectopic expression of Pax4 in the mouse pancreas converts progenitor cells into alpha and subsequently beta cells Collombat P, Xu X, Ravassard P, Sosa-Pineda B, Dussaud S, Billestrup N, Madsen OD, Serup P, Heimberg H, and Mansouri,A (2009) Cell 138, 449-62 4. Embryonic endocrine pancreas and mature beta cells acquire alpha and PP cell phenotypes upon Arx misexpression Collombat P, Hecksher-Sørensen J, Krull J, Berger J, Riedel D, Herrera PL, Serup P, Mansouri A (2007) J Clin Invest 117(4), 961-70 5. Opposing actions of Arx and Pax4 in endocrine pancreas development. Collombat P, Mansouri A, Hecksher-Sorensen J, Serup P, Krull J, Gradwohl G, Gruss P. (2003) Genes Dev 17, 2591-603 Awards 2013: Appolinaire Bouchardat Award 2011: ERC Starting Grant 2009: JDRF Career Development Award 2009: Avenir Excellency INSERM 2009: Schlumberger Prize
-
Cristofari Gaël – Retrotransposon and genome plasticity
CRISTOFARI Gaël

CRISTOFARI Gael, Inserm Group Leader IRCAN Gael.Cristofari@unice.fr04 93 37 70 87 website 
Retrotransposon and genome plasticity
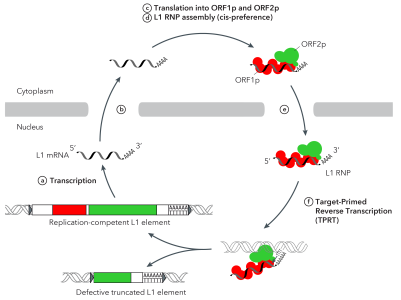 A model for the replication of human L1 retrotransposons
A model for the replication of human L1 retrotransposonsThe abundance of transposable elements in the human genome was one of the most surprising discoveries of genomics. More than 40% of our DNA is made of mobile genetic elements, also called »jumping genes ». Retrotransposons, such as the LINE-1 elements (L1), replicate through a reverse transcription process and are the most abundant active elements in our genome. They play a fundamental role in the current evolution and plasticity of the human genome and can result in the appearance of new genetic diseases or in tumorigenesis. They are repressed in most somatic cells in the adult. However, very recent genome-wide sequencing studies have revealed extensive retrotransposition in several epithelial cancer types (colorectal, lung, ovarian & prostate cancers) but none in glioblastoma or myeloma, suggesting that all tumors are not equal with respects to retrotransposon activation and that transformation or tissue-specific pathways might influence the extent of their mobilization. Our lab explores the link between retrotransposon mobility and tumor genome dynamics and instability. We also aim at understanding how the retrotransposition machinery is regulated at the molecular and cellular levels. To address these goals, we combine biochemistry, molecular & cellular biology, genomics and bioinformatics. We work in close collaboration with clinicians to get access to tumor samples. Selected Publications
1. The specificity and flexibility of l1 reverse transcription priming at imperfect T-tracts. Monot C, Kuciak M, Viollet S, Mir AA, Gabus C, Darlix JL, Cristofari G. (2013) PLoS Genet 9, e1003499 2. Structure of active dimeric human telomerase. Sauerwald A, Sandin S, Cristofari G, Scheres SH, Lingner J, Rhodes D. (2013) Nat Struct Mol Biol 20, 454-60 3. RNA-mediated interference and reverse transcription control the persistence of RNA viruses in the insect model Drosophila. Goic B, Vodovar N, Mondotte JA, Monot C, Frangeul L, Blanc H, Gausson V, Vera-Otarola J, Cristofari G, Saleh MC. (2013) Nat Immunol. 14, 396-403 4. Human telomerase RNA accumulation in Cajal bodies facilitates telomerase recruitment to telomeres and telomere elongation. Cristofari G, Adolf E, Reichenbach P, Sikora K, Terns RM, Terns MP, Lingner J. (2007) Mol Cell. 27, 882-889 5. A 5’-3’ long-range interaction in Ty1 RNA controls its reverse transcription and retrotransposition. Cristofari G, Bampi C, Wilhelm M, Wilhelm FX, Darlix JL. (2002) EMBO J. 21, 4368-79 Awards 2009: Laureate of the European Research Council (ERC Starting Grant) 2009: Laureate of the French National Academy of Medicine (’Albert Sézary’ Prize) 2008: Awardee of the AVENIR program 2003-2004: EMBO long-term fellow 2003: Laureate of the ‘Jacques Piraud’ Prize, French Foundation for Medical Research (FRM)
-
Dani Christian – Stem cells and differentiation
DANI Christian

DANI Christian, Inserm Group Leader iBV dani@unice.fr 04 93 37 76 47 http://ibv.unice.fr/EN/equipe/dani.php 
Stem cells and differentiation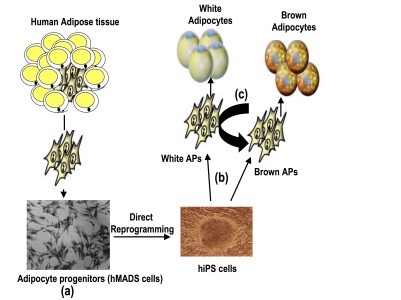 Illustration of cell models established in the team and the main questions we are addressing.
Illustration of cell models established in the team and the main questions we are addressing.While there has been major progress in defining the transcriptional networks that control terminal differentiation of white and brown adipocyte progenitors (APs), much less is known regarding the developmental steps preceding AP generation in mouse, and nothing is known in humans. We propose to identify signalling pathways and molecular events that regulate the earliest events of brown and white adipocyte generation during differentiation of human induced pluripotent stem (hiPS) cells. We have recently shown that retinoic acid (RA) pathway activation promotes white adipocyte generation, whereas it inhibits the brown adipocyte lineage. In contrast, the TGFβ pathway is required for brown adipocyte generation. Based on these findings, we have developed a procedure to selectively derive brown and white APs. We propose to characterize them at a molecular level to identify regulators of AP fate. Selected Publications
1. Transplantation of a multipotent cell population from human adipose tissue induces dystrophin expression in the immunocompetent mdx mouse. Rodriguez AM, Pisani D, Dechesne CA, Turc-Carel C, Kurzenne JY, Wdziekonski B, Villageois A, Bagnis C, Breittmayer JP, Groux H, Ailhaud G, and Dani C. (2005) J Exp Med May 2;201(9), :1397-405. 2. Autocrine FGF2 signaling is critical for self-renewal of Human Multipotent Adipose-Derived Stem Cells Zaragosi LE, Ailhaud G, and Dani C (2006) Stem Cells 24(11), 2412-9 3. Human Multipotent Adipose-derived Stem Cells Differentiate into Functional Brown Adipocytes Elabd C, Chiellini C, Carmona M, Galitzky J, Cochet O, Petersen R, Penicaud L, Kristiansen K, Bouloumie A, Casteilla L, Dani C, Ailhaud G, and Amri EZ (2009) Stem Cells 27(11), 2753-60 4. Activin A plays a critical role in proliferation and differenrentiation of human adipose progenitors Zaragosi LE, Wdziekonski B, Villageois P, Keophiphath M, Maumus M, Tchkonia T, Bourlier V, Mohsen-Kanson T, Ladoux A, Elabd C, Scheideler M, Trajanoski Z, Takashima Y, Amri EZ, Lacasa D, Sengenes C, Ailhaud G, Clément K, Bouloumie A, Kirkland JL , and Dani C (2010) Diabetes 56, 2513-2521 5. Small RNA sequencing reveals miR-642a-3p as a novel adipocyte-specific microRNA and miR-30 as a key regulator of human adipogenesis Zaragosi LE, Wdziekonski B, Le Brigand K, Villageois P, Mari B, Waldmann R, Dani C, and Barbry P. (2011) Genome Biol. 18, 12(7):R64.
-
Delaunay Franck – Circadian System Biology
DELAUNAY Franck

DELAUNAY Franck, UNS Group Leader iBV delaunay@unice.fr 04 92 07 68 38 ibv.unice.fr/EN/equipe/delaunay.php 
Circadian System Biology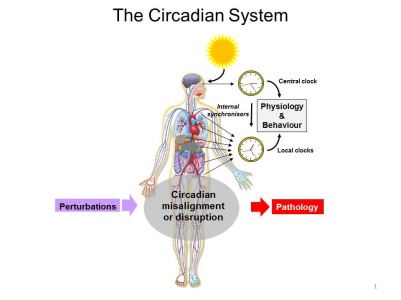 Current model of the mammalian circadian timing system.
Current model of the mammalian circadian timing system.Most organisms show daily rhythms in physiology and behaviour. These rhythms are driven endogenous clocks oscillating with a circadian (~ 24 h) period and synchronized with the external light/dark cycle. In mammals, circadian clocks are organized in a hierarchical and highly integrated system comprising a central clock in the brain that synchronises local clocks throughout the periphery. The clock-controlled transcriptional programme is highly tissue-specific and nearly all key biological processes appear to be linked to the circadian timing system, including for instance the sleep/wake cycle, core body temperature, hormone secretion, metabolism, blood pressure, cell division, and immune function. Consequently, circadian disruption or misalignment is increasingly recognized as an important factor in the context of major diseases including cancer and metabolic disorders. Our aim is to understand how circadian clocks regulate physiological outputs in mammals and reciprocally, determine the mechanisms whereby key biological processes influence the circadian timing system. We currently investigate the interactions between core clock genes and energy metabolism with a focus on the role of specific transcriptional regulators. A second line of research is dedicated to the analysis of the coupling between the circadian clock and cell cycle oscillators in normal and cancer cells. Selected Publications
1. The nuclear receptor REV-ERBalpha is required for the daily balance of carbohydrate and lipid metabolism Delezie J, Dumont S, Dardente H, Oudart H, Grechez-Cassiau A, Klosen P, Teboul M, Delaunay F, Pevet P, and Challet E. (2012) FASEB Journal 26, 3321-3335 2. Kruppel-like factor KLF10 is a link between the circadian clock and metabolism in liver Guillaumond F, Grechez-Cassiau A, Subramaniam M, Brangolo S, Peteri-Brunback B, Staels B, Fievet C, Spelsberg TC, Delaunay ., and Teboul M (2010) Mol Cell Biol 30, 3059-3070 3. Cancer inhibition through circadian reprogramming of tumor transcriptome with meal timing. Li XM, Delaunay F, Dulong S, Claustrat B, Zampera S, Fujii Y, Teboul, M, Beau, J, and Levi F (2010) Cancer Research 70, 3351-3360 4. The circadian clock component BMAL1 is a critical regulator of p21WAF1/CIP1 expression and hepatocyte proliferation Gréchez-Cassiau A, Rayet B, Guillaumond F, Teboul M, Delaunay F (2008) J Biol Chem 238, 4535-4541 Awards 2001: ACI Jeune Chercheur
-
Descombes Xavier – Computational Morphometry and Morphodynamic of Cellular & Supracellular Structures (MORPHEME team : Inria/iBV/I3S)
DESCOMBES Xavier

DESCOMBES Xavier, Inria Group Leader Inria Xavier.Descombes@inria.fr 0492942728 http://www-sop.inria.fr/morpheme/ 
Computational Morphometry and Morphodynamic of Cellular & Supracellular Structures
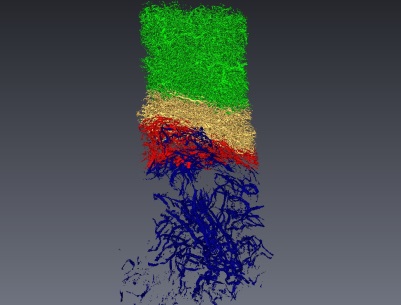
Vascular network segmentation from X-ray micro-tomography : Necrosis (blue), Tumor (red), Tumor Periphery (yellow) and Sane Tissue (green) The scientific objectives of MORPHEME are to characterize and model the development and the morphological properties of biological structures from the cell to the supra-cellular scale. Being at the interface between computational science and biology, we plan to understand the morphological changes that occur during development combining in vivo imaging, image processing and computational modelling. Selected Publications
1. Amplitude-based data selection for optimal retrospective reconstruction in micro-SPECT Breuilly M, Malandain G, Guglielmi J, Marsault R, Pourcher T, Franken P and Darcourt J. (2013) Physics in Medicine and Biology 58(8), 26-57 2. Sparse Poisson Noisy Image Deblurring Carlavan M and Blanc-Féraud L. (2012) IEEE Transactions on Image Processing 21(4), 1834-1846 3. Trends in Bio Imaging and Signal Processing Olivo-Marin JC, Blanc-Feraud L, Unser M, Laine A and Lelieveldt B. (2011) IEEE Signal Processing Magazine ., . 4. Axon Extraction from Fluorescent Confocaql Microscopy Images Mottini A, Descombes X and Besse F. (2012) ISBI – International Symposium on Biomedical Imaging , 5. Automatic Dendrite Spines Detection from X-Ray Tomography Volumes Descombes X, Malandain G, Fonta C, Negyessy L and Mosko R. (2013) ISBI – International Symposium on Biomedical Imaging , Awards 2008: Prix de la Recherche – Human Health category
-
Feral Chloé – Epithelial homeostasis and tumorigenesis
FERAL Chloé

FERAL Chloe, Inserm Group Leader IRCAN chloe.feral@inserm.fr 04 93 37 76 93 Website 
Epithelial homeostasis and tumorigenesis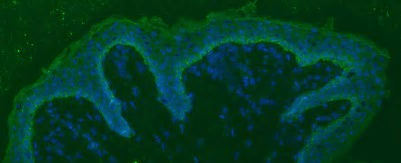 CD98hc Expression in the basal layer of human epidermis (green CD98hc, blue DAPI)
CD98hc Expression in the basal layer of human epidermis (green CD98hc, blue DAPI)The cells that compose the epithelium are constantly renewed and they rest on a basal membrane composed of highly organized network of extracellular matrix (ECM) proteins. To maintain homeostasis in an adult tissue, cell proliferation must be tightly regulated, yet how this is controlled is not fully understood. When this balance is lost epithelial cancers (or carcinomas) arise. In particular, squamous cell carcinoma (SCC) is the second most common type of skin cancer and its incidence increases with age. Our lab is investigating the role of CD98hc, a dual function transmembrane protein, in epidermal tumor formation.CD98 (4F2), is highly expressed in epithelium, which is a heterodimer of a common heavy chain (CD98hc, SLC3A2), and an acid transporter subunit. CD98hc also interacts with ECM receptors, integrins. Thus, via these interaction, CD98hc has two main functions: amino acid transporter and integrin signaling modulator.Our work aims at determining how CD98 interactions contribute to epithelial homeostasis and tumorigenesis. Selected Publications
1. CD98hc (SLC3A2) Regulation of Skin Homeostasis Wanes with Age. Boulter E, Estrach S, Errante A, Pons C, Cailleteau L, Tissot F, Meneguzzi G, Féral CC. (2013) J Exp Med. 210(1), 173-90. 2. Dependence of proliferative vascular smooth muscle cells on CD98hc (4F2hc, SLC3A2). Fogelstrand P, Féral CC, Zargham R, Ginsberg MH. (2009) J Exp Med. 206(11), 2397-406. 3. CD98hc (SLC3A2) participates in fibronectin matrix assembly by mediating integrin signaling. Féral CC, Zijlstra A, Tkachenko E, Prager G, Gardel ML, Slepak M, Ginsberg MH. (2007) J Cell Biol. 178(4), 701-11 4. CD98hc (SLC3A2) mediates integrin signaling. Feral CC, Nishiya N, Fenczik CA, Stuhlmann H, Slepak M, Ginsberg MH. (2005) Proc Natl Acad Sci U S A 102(2), 355-60 Awards 2009-2013: Avenir
-
Frendo Pierre – Nitrogen-fixing symbiosis and redox state
FRENDO Pierre

FRENDO Pierre, UNS Group Leader ISA frendo@unice.fr0492386638 http://www6.paca.inra.fr/institut-sophia-agrobiotech_eng/Research-teams/SYMBIOSE 
Nitrogen-fixing symbiosis and redox state
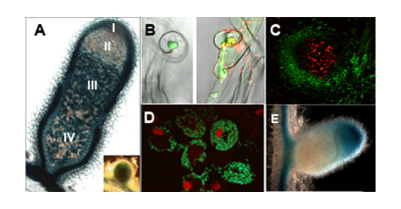 Leguminous – Rhizobium symbiosis : Nodule (A); Detection of NO (B) and of H2O2 (C); bacteroids (D) and gene expression in nodule (E).
Leguminous – Rhizobium symbiosis : Nodule (A); Detection of NO (B) and of H2O2 (C); bacteroids (D) and gene expression in nodule (E).The team focuses on improving our knowledge of plant/nitrogen-fixing bacteria (Rhizobium) by studying the role of the cellular redox state during the symbiotic interaction. Work is focused on two essential steps in the symbiotic relationship: the study of reception mechanisms of the symbiotic partner leading to a functional nodule and the understanding of the nodule senescence phenomenon leading to the breakdown of the symbiotic association. Within this framework, it especially studies three major molecules involved in the regulation of the cellular redox state: hydrogen peroxide (H2O2), nitrogen monoxide (NO) and glutathion (GSH). Selected Publications
1. Hydrogen peroxide and nitric oxide: key regulators of the legume – Rhizobium and mycorrhizal symbioses Puppo A, Pauly N, Boscari A, Mandon K, Brouquisse R. (2013) Antioxid Redox Signal 18, 2202-2219 2. Expression Dynamics of the Medicago truncatula Transcriptome during the Symbiotic Interaction with Sinorhizobium meliloti: Which Role for Nitric Oxide? 161, 425–439. Boscari A, del Giudice J, Ferrarini A, Venturini L, Zaffini AL, Delledonne M, Puppo A. (2013) Plant Physiol. 161, 425-439 3. Peribacteriod space acidification: a marker of mature bacteroid functioning in Medicago truncatula nodules. Pierre O, Engler G, Hopkins J, Brau F, Boncompagni E, and Hérouart D. (2013) Plant Cell Environ Doi: 10.1111/, pce.12116 4. Hydrogen peroxide-regulated genes in the Medicago truncatula– Sinorhizobium meliloti symbiosis. Andrio E, Marino D, Marmeys A, Dunoyer de Segonzac M, Damiani I, Genre A, Huguet S, Frendo P, Puppo A and Pauly N. (2013) New phytol 198, 179-189 5. (homo)glutathione Deficiency Impairs Root-knot. Nematode Development in Medicago truncatula Baldacci-Cresp F, Chang C, Maucourt M, Deborde C, Hopkins J, Lecomte P, Brouquisse R, Moing A, Abad P, Hérouart D, Puppo A, Favery B and Frendo P. (2012) PloS Pathogens 8, e1002471
-
Fürthauer Maximilian – Membrane dynamics and cell signaling in animal development
FURTHAUER Maximilian

FURTHAUER Maximilian, CNRS Group Leader iBV furthauer@unice.fr 0492076439 http://ibv.unice.fr/EN/equipe/furthauer.php 
Membrane dynamics and cell signaling in animal development
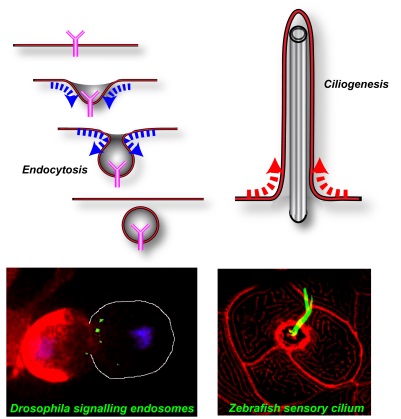 Membrane deformations in endocytosis and ciliogenesis
Membrane deformations in endocytosis and ciliogenesisThe regulation of membrane shape has important consequences for cell signalling: While endocytic invaginations internalize signalling molecules from the cell surface, membrane protrusions probe the cellular environment. Our work at the interface of cellular and developmental biology uses Zebrafish and Drosophila to study how membrane architecture contributes to the regulation of cell signalling in animal development: 1) Membranes are organized into distinct functional domains. Consequently, signalling molecules have to be targeted to precise environments to exert their function. We have developed a novel live imaging approach that allows for the first time to visualize the endocytic transport of endogenous signalling molecules in intact living embryos. Using this approach we are studying how Delta/Notch signalling, one of the major cellular signalling pathways, is regulated during the development of the nervous system. 2) Cilia are specialized membrane protrusions that allow cells to communicate with their environment by chemosensation, mechanosensation and the creation of fluid flows. We have recently uncovered important new functions of intracellular transport proteins in the formation and function of ciliated organs. Presently we are using a combination of genetic, cell biological and live imaging approaches to characterize novel aspect of ciliary organ function. Selected Publications
1. Directional Delta and Notch trafficking in Sara endosomes during asymmetric cell division. Coumailleau F, Fürthauer M, Knoblich JA, and González-Gaitán M. (2009) Nature 458, 1051-1055 2. Endocytic regulation of notch signalling during development. Fürthauer M and González-Gaitán M. (2009) Traffic 10, 792-802 3. Endocytosis, asymmetric cell division, stem cells and cancer: unus pro omnibus, omnes pro uno. Fürthauer M and González-Gaitán M. (2009) Mol Oncol 3, 339-353 4. Fgf signalling controls the dorsoventral patterning of the zebrafish embryo. Fürthauer M, Van Celst J, Thisse C and Thisse,B. (2004) Development 131, 2853-2864 5. Sef is a feedback-induced antagonist of Ras/MAPK-mediated FGF signalling. Fürthauer M, Lin W, Ang SL, Thisse B and Thisse C. (2002) Nat Cell Biol 4, 170-174 Awards 2012 : ARC Projet 2011: HFSP Career Development Award 2010: ATIP/Avenir Junior Group Leeder Programme 2005: HFSP Long Term Postdoctoral Fellowship 2004: EMBO Postdoctoral fellowship
-
Gilson Eric – Telomere, senescence and cancer
GILSON Eric

GILSON Eric, UNS Group Leader IRCAN Eric.Gilson@unice.fr06 07 27 29 73 Website 
Telomere, senescence and canceR
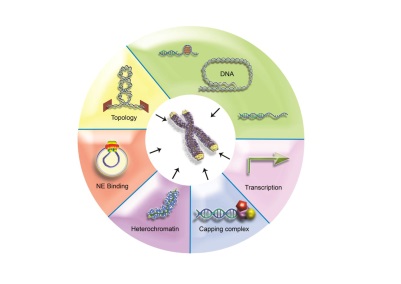 Data from our team indicate that topological stress may constitute a conserved signaling pathway to recruit capping proteins.
Data from our team indicate that topological stress may constitute a conserved signaling pathway to recruit capping proteins.Task #1: Structure and function of of telomeric chromatin in normal and cancer cells 1) The role of nucleosomes in telomere biology We plan to study in human cells : – the telomere chromatin organization by in vitro reconstitution, biochemical analyses and in vivo imaging combining AFM with fluorescence microscopy; – the functional interactions between shelterin components and nucleosomes; – the nature of the epigenetic changes that occurred at the onset of replicative senescence. 2) The role of TRF2 as a DNA topology stress sensor We will measure the topological status of telomeres in living cells and to screen for genes that may be direct actors or participate in the regulation of telomeric topological barrier(s).We will also analyze telomeric replication using an in vitro system.Task #2 Roles of shelterin in long-range chromatin structure and gene expression We will study the mechanism(s) by which extratelomeric TRF2 binding sites regulate the expression of extratelomeric genes.Task # 3 Role of telomeres in oncogenesis We develop three approaches to better understand the role of telomeres in oncogenesis :1) To assay the tumorigenicity of cancer cells expressing separation–of-function mutants of TRF2; 2) Based on our results identifying genes involved in NK cell biology and angiogenesis as TRF2 targets; 3) Based on our results involving IL-6 in telomere capping. Selected Publications
1. TRF2 and apollo cooperate with topoisomerase 2alpha to protect human telomeres from replicative damage Ye J, Lenain C, Bauwens S, Rizzo A, Saint-Leger A, Poulet A, Benarroch D, Magdinier F, Morere J, Amiard S, Britton, P Calsou, B Salles, A Bizard, M Nadal, E Salvati, L Sabatier, Y Wu, A Biroccio, A Londoño-Vallejo, MJ Giraud-Panis and E. Gilson. (2010) Cell 142(2), 230-242 2. A two-step model for senescence triggered by a single critically short telomere Abdallah P, Luciano P, Runge KW, Lisby M, Géli V, Gilson E, Teixeira MT. (2009) Nature Cell biol.11, 988-93 3. TRF2 inhibits a cell-extrinsic pathway through which Natural Killer cells eliminate cancer cells Biroccio AM, Cherfils-Vicini J, Augereau A, Pinte S, Bauwens S, Ye J, Jamet K, Cervera L, Mendez-Bermudez A,Poncet D, Grataroli R, T’kint de Rodenbeeke C, Salvati E, Rizzo A, Zizza P, Ricoul M, Cognet C, Kuilman T, Duret H, Lépinasse F, Marvel J, Verhoeyen E, Cosset FL, Peeper D, Smyth M, Londoño-Vallejo A, Sabatier L, Picco V, Pages G, Scoazec JY, Stoppacciaro A, Leonetti C, Vivier E, Gilson E. (2013) Nature Cell biol in press, na 4. Telomere protection and TRF2 expression are enhanced by the canonical Wnt signalling pathway Diala I, Wagner N, Magdinier F, Shkreli M, Sirakov M, Bauwens S, Schluth-Bolard C, Simonet T, Renault VM, Ye J, Djerbi A, Pineau P, Choi J, Artandi S, Dejean A, Plateroti M, Gilson E.(2013) EMBOr 14, 356-63 5. The human TTAGGG Repeat Factors 1 and 2 bind to a subset of interstitial telomeric sequences and satellite repeats T Simonet, LE Zaragosi, C Philippe, K Lebrigand, C Schouteden, A Augereau, S Bauwens, J Ye, M Santagostino, E Giulotto, F Magdinier, B Horard, P Barbry, R Waldmann, and E Gilson . (2011) Cell research 21, 1028-38 Awards 2010: Prize EUROCANCER 2010: Prize Allianz-Institut de france 2003: Elected EMBO member 2003: Prize Marguerite Delahautemaison of “Fondation de la Recherche Médicale”
-
Glaichenhaus Nicolas – Immunology and immune tolerance
GLAICHENHAUS Nicolas

GLAICHENHAUS Nicolas, UNS Group Leader IPMC nicolas.glaichenhaus@unice.fr 0493957785 http://www.ipmc.cnrs.fr 
Immunology and immune tolerance
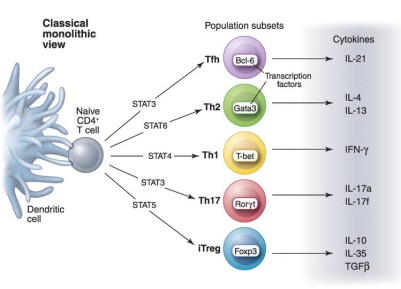 Helper T cell differentiation (O’Shea and Paul. 2010. Science)
Helper T cell differentiation (O’Shea and Paul. 2010. Science)Our team has a broad interest in immunology, and more specifically on T cell differentiation, antigen presentation and immune tolerance. Our current specific aims are (1) to identify the molecular and cellular mechanisms by which the environment perceived by brain influences immune response to foreign antigens or to cancer cells, (2) to elucidate the role of chemokine and chemokine receptors in airway inflammation and (3) to decipher the mechanisms by which viruses could exacerbate airway inflammation in individuals with allergic asthma. Selected Publications
1. Langerin+ dendritic cells are responsible for LPS-induced reactivation of allergen-specific Th2 responses in postasthmatic mice Ortiz-Stern A, Kanda A, Mionnet C, Cazareth J, Lazzari A, Fleury S, Dombrowicz D, Glaichenhaus N, Julia V (2011) Mucosal Immunol 4, 343-53 2. The oral administration of bacterial extracts prevents asthma via the recruitment of regulatory T cells to the airways Navarro S, Cossalter G, Chiavaroli C, Kanda A, Fleury S, Lazzari, A. Cazareth J, Sparwasser T, Dombrowicz D, Glaichenhaus N, Julia V (2011) Mucosal Immunol 4, 53-65 3. Direct visualization of peptide/MHC complexes at the surface and in the intracellular compartments of cells infected in vivo by Leishmania major Muraille E, Gounon P, Cazareth J, Hoebeke J, Lippuner C, Davalos-Misslitz A, Aebischer T, Muller S, Glaichenhaus N, Mougneau E (2010) PLoS Pathog 6, e1001154 4. Breast milk immune complexes are potent inducers of oral tolerance in neonates and prevent asthma development Mosconi E, Rekima A, Seitz-Polski B, Kanda A, Fleury S, Tissandie, E. Monteiro R, Dombrowicz D, Julia V, Glaichenhaus N, Verhasselt V (2010) Mucosal Immunol 3, 461-474 5. CX3CR1 is required for airway inflammation by promoting T helper cell survival and maintenance in inflamed lung Mionnet C, Buatois V, Kanda A, Milcent V, Fleury S, Lair D, Langelot M, Lacoeuille, Y. Hessel E, Coffman R, Magnan A, Dombrowicz D, Glaichenhaus N, Julia V (2010) Nat Med 16, 1305-1312 Awards 2010: Elected senior member of the Institut Universitaire de France (IUF) 1998: Elected senior member of the European Molecular Biology Organisation (EMBO)
-
Gouzé Jean-Luc – Biological Control of artificial ecosystems (BIOCORE team: INRIA/INRA/CNRS/UPMC)
GOUZE Jean-Luc

GOUZE Jean-Luc, Inria Group Leader Inria jean-luc.gouze@inria.fr0492387875 https://team.inria.fr/biocore/ 
Biological Control of artificial ecosystems (BIOCORE team: INRIA/INRA/CNRS/UPMC)
Genetic Network Analyzer(de Jong et al.)
We build and study mathematical dynamical models of biological systems: intracellular models (of genetic and/or metabolic type : genetic networks, metabolic networks, signalling networks ), models of populations or at the scale of the ecosystems. Our tools are dynamical systems and control theory.These models are qualitative or quantitative. Parameters are then fitted with experimental data. Selected Publications
1. A theoretical exploration of birhythmicity in the p53-Mdm2 network Abou-Jaoudé W, Chaves M. and Gouzé J.-L. (2011) PLOS one 6(2), e17075 2. Oscillations induced by different timescales in signal transduction modules regulated by slowly evolving protein–protein interactions Ndiaye, I and Chaves, M and Gouzé, J-L (2010) IET systems biology 4 (4), 263-276 3. Robustness and fragility of Boolean models for genetic regulatory networks M. Chaves, R. Albert and E.D. Sontag. (2005) J. Theoretical Biology 235 (3), 431-449 4. Qualitative simulation of genetic regulatory networks using piecewise-linear models De Jong, H. , Gouzé J-L, Hernandez, C, Page, M., Sari, T. and Geiselmann, J (2004) Bulletin of mathematical biology 66 (2), 301-340
-
Tran Albert/Gual Philippe – Hepatic complications in obesity
GUAL Philippe/TRAN Albert

GUAL Philippe, Inserm Group Leader C3M gual@unice.fr04 89 06 42 23 http://www.unice.fr/c3m/EN/Equipe8.html 
TRAN Albert, Inserm Group Leader C3M albert.TRAN@unice.fr 0492035943 http://www.unice.fr/c3m/EN/Equipe8.html 
Hepatic complications in obesity
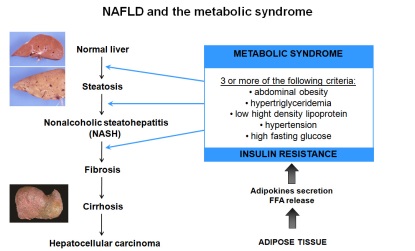 Hepatic complications in obesity
Hepatic complications in obesityThe hepatic complications of obesity, one of the most common forms of chronic liver diseases. range from steatosis to steatohepatitis (Non Alcoholic SteatoHepatitis, NASH), fibrosis, cirrhosis and finally hepatocellular carcinoma. The diagnosis of NASH requires a liver biopsy. The pathophysiological mechanisms of the progression of these diseases are complex, including insulin-resistance, cellular stress and upregulation of pro-inflammatory factors. These factors can originate from intra- or extra-hepatic sites, particularly the adipose tissue. Our main objectives are: 1) To develop a non invasive index from clinical data and the identified markers allowing the diagnosis of NASH in our cohort of 800 obese patients, for whom serum and hepatic and adipose tissue biopsies have been obtained. 2) To study the role of identified and new players in the progression of liver complications. This will be evaluated in different cellular and animal models. 3) To study the development of the liver complications in achondroplasia in animal models. 4) To evaluate the effect of pharmacological and chirurgical treatment on prevention and correction of liver complications in animal models.The results of the present project should bring new insights in the understanding of the mechanistic of hepatic complications in obesity and to propose better diagnostic and therapeutic approaches. Selected Publications
1. Regular coffee but not Espresso drinking is protective against fibrosis in a cohort mainly composed of morbidly obese European women patients with NAFLD undergoing bariatric surgery. Anty R, Marjoux S, Iannelli A, Patouraux S, Schneck AS, Bonnafous S, Gire C, Amzolini A, Ben-Amor I, Saint-Paul MC, Mariné-Barjoan E, Pariente A, Gugenheim J, Gual P, Tran A. (2012) J of Hepatology 57, 1090-6. 2. Identification of Adipose Tissue Dendritic Cells Correlated With Obesity-Associated Insulin-Resistance and Inducing Th17 Responses in Mice and Patients. Bertola A*, Ciucci T*, (*co-first authors) Rousseau D, Bourlier V, Duffaut C, Bonnafous S, Blin-Wakkach C, Anty R, Iannelli A, Gugenheim J, Tran A, Bouloumié A, Gual P$, Wakkach A$ ($ co-last authors). (2012) Diabetes 61, 2238-47 4. Elevated expression of osteopontin may be related to adipose tissue macrophage accumulation and liver steatosis in morbid obesity. Bertola, A., Deveaux V, Bonnafous S, Rousseau D, Anty R, Wakkach A, Dahman M, Tordjman J, Clement K, McQuaid SE, Frayn KN, Huet PM, Gugenheim J, Lotersztajn S, Le Marchand-Brustel Y, Tran A, and Gual P (2009) Diabetes 58, 125-33 5. Increased adipose tissue expression of hepcidin in severe obesity is independent from diabetes and NASH. Bekri, S*, Gual P* (Co first authors), Anty R, Luciani N, Dahman M, Ramesh B, Iannelli A, Staccini-Myx A, Casanova D, Ben Amor I, Saint-Paul MC, Huet PM, Sadoul JL, Gugenheim J, Srai SK, Tran A and Le Marchand-Brustel Y. (2006) Gastroenterology 131, 788-96 Awards 2003 : Prix de Recherche Clinique Insitut Roche de l’obésité « Rôle de l’IL6 dans les complications de l’obésité ». P. Gual
-
Hofman Paul – Carcinogenesis related chronic active inflammation
HOFMAN Paul

HOFMAN Paul, Inserm Group Leader IRCAN hofman.p@chu-nice.fr 06 17 01 27 54 www.biobank06.com 
Carcinogenesis related chronic active inflammation
 Crosstalk between neutrophil and lung cancer tumor
Crosstalk between neutrophil and lung cancer tumorEpidemiological and histological data highlight that an intense and repeated neutrophil infiltration over a long period time is strongly linked to a high risk of carcinoma onset. However, even though different mechanisms claim to explain the onset of carcinogenesis in response to repeated neutrophil transepithelial migration and/or neutrophil epithelial contact. The pathophysiological of this process is complex and its understanding remains obscure. Within a tumour, cancer cells are surrounded by an inflammatory microenvironment containing different cell subtypes including polymorphonuclear leukocytes (PMNL, i.e. neutrophils). Accumulating evidence strongly suggests that cancer cells attract inflammatory cells, in particular PMNL, and might subvert their function to promote tumour cell proliferation, resistance again cell death and metastasis. However, the relationship between cytokines, epithelial cells and PMNL, and the consequence of such cell-cell interactions on regulation of downstream events (in particular, miRNA regulation and/or protein expression) has been poorly investigated, in particular during the critical transition from a chronic “active” inflammatory lesion to a carcinoma.Our project is divided into the following objectives: i) to characterize the role of the microenvironment on PMNL maturation (aim 1), ii) to characterize the involvement of PMNL in tumour initiation (aim 2), and in tumour progression and dissemination (aim 3). Selected Publications
1. Diagnostic value of immunohistochemistry for the detection of the BRAFV600E mutation in primary lung adenocarcinoma Caucasian patients. Ilie M, Long E, Hofman V, Dadone B, Marquette CH, Mouroux J, Vignaud JM, Begueret H, Merlio JP, Capper D, von Deimling A, Emile JF, Hofman P. (2013) Ann Oncol 24, 742-748 2. ALK-gene rearrangement: a comparative analysis on circulating tumour cells and tumour tissue from patients with lung adenocarcinoma. Ilie M, Long E, Butori C, Hofman V, Coelle C, Mauro V, Zahaf K, Marquette CH, Mouroux J, Paterlini-Bréchot P, Hofman P (2012) Ann Oncol 23, 2907-2911 3. Predictive clinical outcome of the intratumoral CD66b-positive neutrophil-to-CD8-positive T-cell ratio in patients with resectable nonsmall cell lung cancer. Ilie M, Hofman V, Ortholan C, Bonnetaud C, Coëlle C, Mouroux J and Hofman P. (2012) Cancer 118, 1726-1737 4. A synonymous variant in IRGM alters a binding site for miR-196 and causes deregulation of IRGM-dependent xenophagy in Crohn’s disease. Brest P, Lapaquette P, Souidi M, Lebrigand K, Cesaro A, Vouret-Craviari V, Mari B, Barbry P, Mosnier JF, Hébuterne X, Harel-Bellan A, Mograbi B, Darfeuille-Michaud A, Hofman P (2011) Nat Genet 43, 242-254 5. Preoperative circulating tumor cell detection using the isolation by size of epithelial tumor cell method for patients with lung cancer is a new prognostic biomarker. Hofman V, Bonnetaud C, Ilie MI, Vielh P, Vignaud JM, Fléjou JF, Lantuejoul S, Piaton E, Mourad N, Butori C, Selva E, Poudenx M, Sibon S, Kelhef S, Vénissac N, Jais JP, Mouroux J, Molina TJ, Hofman P (2011) Clin Cancer Res 17, 827-835 Awards 2012: Shangai Clinical Cancer Center Prize 2012
-
Hueber Anne-Odile – Death receptors signaling and cancer therapy
HUEBER Anne-Odile

HUEBER Anne-Odile, Inserm Group Leader iBV hueber@unice.fr 0402076447 http://ibv.unice.fr/EN/equipe/hueber.php 
Death receptors signaling and cancer therapy
 Balance between life and death in colorectal cancer cell line
Balance between life and death in colorectal cancer cell lineAlterations in the control of cell survival and death contribute to the pathogenesis of many human diseases. In particular, the dysfunction in the signaling cascade leading to the programmed cell death has been shown a key factor in the development of many types of cancer. Since the last ten years the lab is focus on the understanding of the role of the one of this apoptotic pathways, the one triggered by Fas upon its engagement by its ligand (FasL). Results in our laboratory show that triggering Fas signaling by sublethal doses of FasL can activate proteins with known survival functions (Akt and MAPK), and induce colorectal cancer (CRC) cell proliferation. Also, the evidence of Fas/FasL contribution in CRC metastasis is emerging. A multifaceted scenario has also been presented where CRC cells not only employ the anti-apoptotic and proliferative capacity of the Fas/FasL signaling for local growth but also utilize FasL to induce apotosis in hepatocytes to facilitate the liver metastasis. In addition, invasion of CRC cells and liver metastasis promoted by activating Fas with FasL have been shown dependent on oncogenic K-Ras. This thus links the Fas/FasL non-apoptotic signaling to major colorectal carcinogenesis pathways. Despite the fact that Fas signaling plasticity is central to the balance of life and death decision of the cell, the regulation of this versatility remains obscure. The PhD project will be part of the general focus on the lab: the understanding at a molecular level the versatility of Fas and its importance in the tumoral initiation and progression processes and thus the development of effective CRC therapies. Selected Publications
1. Vesicles released by activated T cells induce both Fas-mediated RIP-dependent apoptotic and Fas-independent non-apoptotic cell deaths Koncz, G., Hancz, A., Chakrabandhu, K., Gogolak, P., Kerekes, K., Rajnavolgyi, E. and Hueber, AO. (2012) Journal of Immunology 189(6), 2815-2823 2. Palmitoylation of the TRAIL receptor DR4 confers an efficient TRAIL-induced cell death signaling Rossin, A., Derouet, M, Abdel Sater, F; and Hueber, A-O. (2010) Biochemical J. 419(1), 185-194. 3. The Extracellular Juxtamembrane Motif of Fas is Required for Fas-Glycosphingolipid Interaction and Fas-induced Cell Death Chakrabandhu, K., Huault, S., Garmy, N., Stebbe, E., Mailfert, S., Marguet, D., Fantini, J, and Hueber, A-O (2008) Cell Death&Differentiation 15(12), 1824-1837 4. The CD95 receptor: apoptosis revisited Marcus E. Peter, Ralph C. Budd, Julie Desbarats, Stephen M. Hedrick, Anne-Odile Hueber, M. Karen Newell, Laurie B. Owen, Richard M. Pope, Juerg Tschopp, Harald Wajant, David Wallach, Robert H., Wiltrout, Martin Zörnig, and David H. Lynch (2007) Cell 129(3), 447-450 5. Fas palmitoylation is required for efficient Fas-induced cell death K. Chabrandhu#, Z. Hérincs#, S. Huault, D. Britta, P. Ling, F. Conchonnaud, D. Marguet, H-T He, and A-O Hueber (2007) The EMBO J. 26(1), 209-220 Awards 2002: Cancerology Price Raymond Rosen
-
Lalli Enzo – Regulatory mechanisms of gene expression in physiopathology
LALLI Enzo

LALLI Enzo, Inserm Group Leader IPMC ninino@ipmc.cnrs.fr 0493957755 www.ipmc.cnrs.fr 
Regulatory mechanisms of gene expression in physiopathology Localization of activated mTOR (red) in the midbody of telophase adrenocortical cancer mitotic cells. Green, beta-tubulin; blue, DAPI.
Localization of activated mTOR (red) in the midbody of telophase adrenocortical cancer mitotic cells. Green, beta-tubulin; blue, DAPI.We use an approach that integrates molecular and cell biology, genomics, animal models and clinical investigations to advance our understanding of the mechanisms of gene expression regulation in health and disease, with a special focus in the development, function and pathology of the adrenal cortex. Selected Publications
1. Impact of neonatal screening and surveillance for the TP53 R337H mutation on early detection of childhood adrenocortical tumors. Custódio G, Parise GA, Kiesel FN, Komechen H, Sabbaga CC, Rosati R, Grisa L, Parise IZS, Pianovski MAD, Fiori CMCM, Ledesma JA, Barbosa JRS, Figueiredo FRO, Sade ER, Ibañez H, Arram SBI, Stinghen ST, Mengarelli LR, Figueiredo MMO, Carvalho DC, Avilla SGA, Woiski TD, Poncio LC, Lima GFR, Pontarolo R, Lalli E, Zhou Y, Zambetti GP, Ribeiro RC, Figueiredo BC. (2013) J Clin Oncol in press 2. Dkk3 is a component of the genetic circuitry regulating aldosterone biosynthesis in the adrenal cortex. El Wakil A, Bandulik S, Guy N, Bendahhou S, Zennaro M-C, Niehrs C, Mari B, Warth R, Barhanin J, Lalli E. (2012) Hum Mol Genet (cover capture) 21, 4922-4929 3. Increased Steroidogenic Factor-1 dosage triggers adrenocortical cell proliferation and cancer. ) Doghman M, Karpova T, Rodrigues GA, Arhatte M, De Moura J, Cavalli LR, Virolle V, Barbry P, Zambetti GP, Figueiredo BC, Heckert LL, Lalli E. (2007) Mol Endocrinol 21, 2968-2987 4. X-linked adrenal hypoplasia congenita is caused by abnormal nuclear localization of the DAX-1 protein. Lehmann SG*, Lalli E*, Sassone-Corsi P. (*Equal contribution) (2002) Proc Natl Acad Sci USA (cover capture) 99, 8225-8230 5. DNA binding and transcriptional repression by DAX-1 blocks steroidogenesis. Zazopoulos E*, Lalli E*, Stocco DM, Sassone-Corsi P. (*Equal contribution) (1997) Nature 390, 311-315 Awards 2008: »Cancer » prize of the French Academy of Medicine 2005: »Jayle » prize of the French Academy of Sciences
-
Lambeau Gérard – Molecular physiopathology of phospholipases A2 and their mediators
LAMBEAU Gérard

LAMBEAU Gérard, CNRS Group Leader IPMC lambeau@ipmc.cnrs.fr 0493957733 www.ipmc.cnrs.fr 
Molecular physiopathology of phospholipases A2 and their mediators
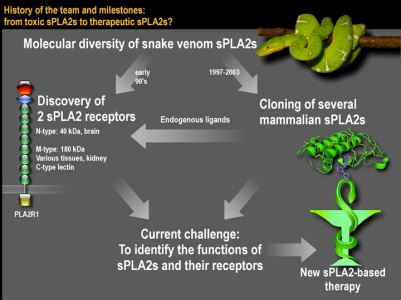 Milestones of the team and current challenges
Milestones of the team and current challengesOur team works on secreted phospholipases A2 (PLA2s), an emerging family of enzymes that hydrolyze phospholipids. Our recent work has led to the identification of novel biological functions of sPLA2s in both physiological and disease conditions including reproduction, inflammation, atherosclerosis, cancer and a rare human kidney disease.Our current objective is to further depict the biological roles and mechanisms of action of sPLA2 in the above biological events and to identify novel functions for sPLA2s by using a combination of in vitro and in vivo experimental approaches. We are also working on the molecular properties of sPLA2s and PLA2R1 and the discovery of novel sPLA2 inhibitors using a combined approach based on biochemistry, bioinformatics and structural biology. Our work may pave the way to novel therapeutic avenues by targeting sPLA2s. Selected Publications
1. Group X secreted phospholipase A2 limits the development of atherosclerosis in LDL receptor-null mice Ait-Oufella H, Herbin O, Lahoute C, Coatrieux C, Loyer X, Joffre J, Laurans L, Ramkhelawon R, Blanc-Brude O, Karabina SA, Girard CA, Payré C, Yamamoto K, Binder CJ, Murakami M, Tedgui A, Lambeau G.(co-last and corresponding author), Mallat Z. (2013) Arterioscler. Thromb. Vasc. Biol. 33, 466-473 2. Group X secreted phospholipase A2 proenzyme is matured by a furin-like proprotein convertase and releases arachidonic acid inside of human HEK293 cells Jemel I, Ii H, Oslund RC, Payre C, Dabert-Gay AS, Douguet D, Chargui K, Scarzello S, Gelb MH, Lambeau G (2011) J. Biol. Chem. 286, 36509-36521 3. Group X phospholipase A2 is released during sperm acrosome reaction and controls fertility outcome in mice Escoffier J, Jemel I, Tanemoto, A, Taketomi Y, Payré C, Coatrieux C, Sato H, Yamamoto K, Masuda S, Pernet-Gallay K, Pierre V, Hara S, Murakami M, De Waard M, Lambeau G (co-last author)Arnoult C (2010) J. Clin. Invest. 120, 1415-1428 4. M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy Beck LH Jr, Bonegio RG, Lambeau G, Beck DM, Powell DW, Cummins TD, Klein JB, Salant DJ (2009) N. Engl. J. Med. 361, 11-21 5. Biochemistry and Physiology of Mammalian Secreted Phospholipases A2 Lambeau G (co-last and corresponding author) and Gelb MH (2008) Annu. Rev. Biochem. 77, 495-520 Awards 2013: Louisiana State University Neuroscience Center of Excellence Award Lecture 2006: Pierre Desnuelle Prize Award of the French Academy of Sciences, Molecular and Cellular Biology section 1995: CNRS Bronze Medal
-
Lemichez Emmanuel – Microbial Toxins in host-pathogen interactions
LEMICHEZ Emmanuel

LEMICHEZEmmanuel, Inserm Group Leader C3M lemichez@unice.fr04 89 06 42 61 http://www.unice.fr/c3m/FR/Equipe6.html 
Microbial toxins in host pathogen interactions
 Cellular effects of toxins targeting Rho GTPases
Cellular effects of toxins targeting Rho GTPasesRac1 and the other Rho proteins are small GTPases, which control all actin cytoskeleton dependent processes, such as adhesion, migration, cytokinesis and phagocytosis and are targeted by numerous bacterial virulence factors.Our studies on the CNF1 toxin of uropathogenic E. coli have revealed a new mode of regulation of active Rac1 by ubiquitin-mediated proteasomal degradation. We have also defined the importance of the Rac1 signaling axis in the control of anti-microbial responses in addition to cell migration and cell invasion by pathogenic bacteria. We found that HACE1 catalyzes the ubiquitylation of the active form of Rac1 with major consequences on Rac1-dependent cell signaling and actin reorganization. Previous studies had established that the down-modulation of HACE1 triggers spontaneous late-onset cancer and kidney tumors in children. Our studies are aimed at clarifying the function of HACE1/Rac1 signaling axis in infection and cancer.We are also studying the dynamic of large transcellular tunnels (TEM) in endothelial cells induced by cAMP- and RhoA-targeting toxins. Time-lapse video analysis has revealed that these tunnels open and close in less than 10 minutes, a dynamic controlled by a subset of I-BAR containing proteins (MIM and ABBA) that are endowed with the property to sense the curvature of membranes. We study other proteins involved in TEM dynamics. Selected Publications
1. The E3 ubiquitin-ligase HACE1 catalyzes the ubiquitylation of active Rac1. Torrino S, Visvikis O, Doye A, Boyer L, Stefani C, Munro P, Bertoglio J, Gacon G, Mettouchi A and Lemichez E (2011) Dev Cell 21, 959-65 2. cAMP signaling by anthrax edema toxin induces transendothelial cell tunnels, which are resealed by MIM via Arp2/3-driven actin polymerization Maddugoda MP, Stefani C, Gonzalez-Rodriguez D, Saarikangas J, Torrino S, Janel S, Munro P, Doye A, Prodon F, Aurrand-Lions M, Goossens PL, Lafont F, Bassereau P, Lappalainen P, Brochard F and Lemichez E (2011) Cell Host & Microbe 10, 464-474 3. Pathogen-derived effectors trigger protective immunity via activation of the Rac2 enzyme and the IMD or Rip kinase signaling pathway. Boyer L, Magoc L, Dejardin S, Cappillino M, Paquette N, Hinault C, Charriere GM, Ip WK, Fracchia S, Hennessy E, Erturk-Hasdemir D, Reichhart JM, Silverman N, Lacy-Hulbert A and Stuart LM (2011) Immunity 35, 536-549 4. Escherichia coli producing CNF1 toxin hijacks Tollip to trigger Rac1-dependent cell invasion Visvikis O, Boyer L, Torrino S, Doye A, Lemonnier M, Lorès P, Rolando M, Flatau G, Mettouchi A, Bouvard D, Veiga E, Gacon G, Cossart P and Lemichez E (2011 ) Traffic 12, 579-590 5. Laminin-binding integrins induce Dll4 expression and Notch signaling in endothelial cells. Estrach S, Cailleteau L, Franco CA, Gerhardt H, Stefani C, Lemichez E, Gagnoux-Palacios L, Meneguzzi G and Mettouchi A. (2011) Circ Res 109, 172-182 Awards 1997: Human Frontier Science Program
-
Leopold Pierre – Genetics and Physiology of growth in Drosophila
LEOPOLD Pierre

LEOPOLD Pierre, Inserm Group Leader iBV leopold@unice.fr0492076445 http://ibv.unice.fr/EN/equipe/leopold.php 
Genetics and Physiology of Growth in Drosophila
 Individuals develop and reach final sizes according to both local (genetic) and environmental (for ex. nutrition) cues.
Individuals develop and reach final sizes according to both local (genetic) and environmental (for ex. nutrition) cues.Animal growth is a complex process, linked to the developmental program in order to form adults with proper size and proportions. Genetics is an important determinant of growth. In addition, organisms use adaptative responses allowing modulating the size of individuals according to environmental cues, among which nutrition. Therefore, a sophisticated crosstalk between local and global cues is at play for the ultimate determination of individual size. Our projects use Drosophila Genetics, Physiology and Cell Biology approaches to decipher the coupling between the environment and the developmental program that determines the final size of an organism. Our specific aims are the following: 1- understanding how insulin/IGF signaling is modulated by nutrition, and in particular identifying the humoral connection between the tissues involved in sensing the nutrients and those producing insulin-like peptides. 2- understanding the molecular basis for growth arrest and the coordination between tissue growth and the developmental timing. 3- Understanding the mechanisms allowing nutrient sensing in the brain and identifying the neuronal pathways controlling feeding according to nutritional cues. 4- Understanding the modulation of simple innate behaviors such as temperature or light preference by developmental cues. Selected Publications
1. Secreted peptide Dilp8 coordinates Drosophila tissue growth with developmental timing Colombani, J. Andersen, D. S. Leopold, P. (2012) Science 336, 582-5 2. The steroid hormone Ecdysone controls systemic growth by repressing dMyc function in Drosophila fat cells. Delanoue, R. Slaidina, M. Léopold, P. (2010) Dev. Cell 18, 1012-1021 3. A Drosophila insulin-like peptide promotes growth during nonfeeding states Slaidina, M. Delanoue, R. Gronke, S. Partridge, L. Leopold, P. (2009) Dev. Cell 17, 874-84 4. Remote control of insulin secretion by fat cells in Drosophila Geminard, C. Rulifson, E. J. Leopold, P. (2009) Cell Metab.10, 199-207 5. Drosophila and the genetics of the internal milieu Leopold, P. Perrimon, N. (2007) Nature 450, 186-188 Awards 2011: Research Prize Inserm 2010: ERC Advanced grant Award 2009: French Academy Prize 2008: Elected EMBO Member
-
Liti Gianni – Population genomics and complex traits
LITI Gianni

LITI Gianni, CNRS Group Leader IRCAN gianni.liti@unice.fr04 93 37 76 72 Website 
Population genomics and complex traits
 Different routes from genotype to phenotype. Complementary forward and reverse genetic approaches to understanding cellular traits
Different routes from genotype to phenotype. Complementary forward and reverse genetic approaches to understanding cellular traitsMost human traits, including many diseases, are regulated by multiple interacting quantitative trait loci (QTLs). Dissecting the genetic mechanisms underlying this phenotypic variation is a major challenge. In our lab, we use the budding yeast, S. cerevisiae, as model to dissect the genetic architecture of multiple traits related to ageing and cancer. We are currently co-leading a major resequencing effort that aims to release high quality genomes for 1002 natural S. cerevisiae strains. This large sample size of full genome sequences coupled to high-throughput phenotyping will provide powerful opportunities for genome wide association studies. The PhD candidate will exploit yeast natural variation to investigate cell signaling pathways. For example, we previously found that widespread natural sequence variation in RAS signalling pathways contribute to quantitative differences in ageing and general stress response. We will use population genomics datasets to understand how the RAS signalling pathway vary and become misregulated in some diverged lineages. This project is suitable for a person either with computational biology background or wet lab experience. In conclusion, we aim to elucidate essential aspects of individual variation among yeast strains with the major goal of understanding the genetic mechanisms underlying complex traits and human diseases. Selected Publications
1. High quality de novo sequencing and assembly of the Saccharomyces arboricolus genome 1. Liti G, Nguyen Ba AN, Blythe M, Müller CA, Bergström A, Cubillos FA, Dafhnis-Calas F, Khoshraftar S, Malla S, Mehta N, Siow CC, Warringer J, Moses AM, Louis EJ and Nieduszynski CA (2013) BMC Genomics 14(1), 1-14 2. Advances in quantitative trait analysis in yeast. Liti G and Louis EJ (2012) PLoS Genetics. 8(8), e1002912., 1-7 3. Revealing the genetic structure of a trait by sequencing a population under selection. Parts L, Cubillos FA, Warringer J, Jain K, Salinas F, Bumpstead SJ, Molin M, Zia A, Simpson JT, Quail MA, Moses AM, Louis EJ, Durbin R and Liti G (2011) Genome Research 21(7), 1131-8 4. Assessing the Complex Architecture of polygenic traits in yeast Cubillos FA, Billi E, Zörgö E, Parts L, Fargier P, Omholt S, Blomberg A, Warringer J, Louis EJ and Liti G. (2011) Molecular Ecology 20(7), 1401-13 5. Population genomics of domestic and wild yeasts. Liti G, Carter DM, Moses AM, Warringer J, Parts L, James SA, Davey RP, Roberts IN, Burt A, Koufopanou V, Tsai IJ, Bergman CM, Bensasson D, O’Kelly MJT, van Oudenaarden A, Barton DBH, Bailes E, Nguyen Ba AN, Jones M, Quail MA, Goodhead I, Sims S, Smith F, Blomberg A, Durbin R and Louis EJ (2009) Nature 19; 458(7236), 337-41
-
Luton Frédéric/Franco Michel – Arf proteins, cell morphology and membrane transpor
LUTON Frédéric/FRANCO Michel

LUTON Frédéric, Inserm Group Leader IPMC luton@ipmc.cnrs.fr 0493957770 www.ipmc.cnrs.fr 
FRANCO Michel, CNRS Group Leader IPMC franco@ipmc.cnrs.fr 0493957770 www.ipmc.cnrs.fr 
Arf proteins, cell morphology and membrane transport
 Acini of breast cancer cell line in 3D cell culture system
Acini of breast cancer cell line in 3D cell culture systemA fundamental question in cell biology is to understand how epithelial cells organize collectively as monolayers, multilayers, tubules or acinis to form a large variety of organs such as lungs, intestine, kidney, mammary glands, etc. It is also of interest in the biology of epithelial cancers since primary tumor development is associated with the loss of the epithelial collective organization and the establishment in cell motility and invasive properties. By combining in vitro reconstitution of epithelial organization using 3D cell culture systems, analyses of human tumor specimen and clinical data we are addressing the role of the small G protein Arf6 and its exchange factor EFA6 (Exchange Factor for Arf6) in breast cancer development. We are currently focusing our studies on a particular breast cancer subtype for which no specific treatment is available. Future directions of this project include the characterization of EFA6/Arf6 associated signaling pathways, which are deregulated in breast cancer development, the search for specific pharmacological compounds directed against in-house identified targets and new breast cancer animal models. Selected Publications
1. Arf6 negatively controls the rapid recycling of the beta2 adrenergic receptor Macia, E., Partisani, M., Paleotti, O., Luton, F. and Franco, M. (2012) J. Cell Science 125, 4026-35 2. USP9x-mediated deubiquitination of EFA6 regulates de novo tight junction assembly Theard, D., Labarrade, F., Partisani, M., Milanini, J., Sakagami, H., Fon, E. A.,Wood, S.A., Franco, M., and Luton F. (2010) Embo J. 29, 1499-509 3. EFA6 facilitates the assembly of the tight junction by coordinating an Arf6-dependent and-independent pathway Klein, S., Partisani M., Franco, M. and Luton, F. (2008) J. Biol. Chem. 283, 30129-30138 4. The PH domain of the Arf6-specific exchange factor EFA6 localizes to the plasma membrane by interacting with PI(4,5)P2 and F-actin Macia, E., Partisani M., Favard, C., Mortier, E., Zimmermann, P., Carlier, M.F., Gounon, P., Luton, F. and Franco, M. (2008) J. Biol. Chem. 283, 19836-19844
-
Magnaldo Thierry/Meneguzzi Guerrino – Genetics and physiopathology of epithelial cancers
MAGNALDO Thierry/MENEGUZZI Guerrino

MAGNALDO Thierry, CNRS Group Leader IRCAN tmagnaldo@unice.fr0493377670 http://ircan.org/ 
MENEGUZZI Guerrino, Inserm Group Leader IRCAN Guerrino.Meneguzzi@unice.fr0493377779 http://ircan.org/ 
Genetics and epigenetics in the ephysiopathology of epithelial cancers
 Co-evolution of tumor-stroma crosstalk in cancer
Co-evolution of tumor-stroma crosstalk in cancerEpithelial cancers arise with age, among which, squamous cell carcinomas (SSCs) are by far the most common and generally lethal in human. Invasiveness of SCCs depends on reciprocal signaling between the tumor cells and their microenvironment (figure 1). In primary tumors, cancer cells are immersed in a complex microenvironment network including fibroblasts. Malignant eptithelial secrete cytokines and growth factors that activate fibroblasts towards acquisition of the Carcinoma Associated Fibrolast (CAF) phenotype. In turn, CAF promote carcinoma cells invasion and malignancy. Our main objective is to unravel molecular pathways bridging epithelial to stroma cells in the context of carcinogenesis.By innovative experimental settings of cancer modeling in vitro and vivo, we aim at determining the signaling pathways associated with epithelial carcinogenesis/stroma activation and, subsequently, initiate suitable therapeutic routes. We have developed two complementary system models, based on either i-) healthy skin cells isolated from rare patients suffering from the cancer prone genetic syndrome Xerodema pigmentosum (T. Magnaldo), or ii-) cancer associated fibroblasts and cancer cell lines established from human sporadic tumors (C. Gaggiolli). Selected Publications
1. Preclinical corrective gene transfer in xeroderma pigmentosum human skin stem cells. Warrick E, Garcia M, Chagnoleau C, Chevallier O, Bergoglio V, Sartori D, Mavilio F, Angulo JF, Avril MF, Sarasin A, Larcher F, Del Rio M, Bernerd F, Magnaldo T. 2012 (2012) Mol Ther 20, 798-807 2. PTCH1 +/- dermal fibroblasts isolated from healthy skin of Gorlin syndrome patients exhibit features of carcinoma associated fibroblasts. Overexpression of matrix metalloproteinase 1 in dermal fibroblasts from DNA repair-deficient/cancer-prone xeroderma pigmentosum group C patients. (2008) Oncogene 27, 5223-5232 3. Confluence switch signaling regulates ECM composition and the plasmin proteolytic cascade in keratinocytes Botta A, Delteil F, Mettouchi A, Vieira A, Estrach S, Négroni L, Stefani C, Lemichez E, Meneguzzi G, Gagnoux-Palacios L (2012) J Cell Sci. 125, 4241-4252 4. ROCK and JAK1 signaling cooperate to control actomyosin contractility in tumor cells and stroma Sanz-Moreno V, Gaggioli C, Yeo M, Albrengues J, Wallberg F, Viros A, Hooper S, Mitter R, Féral CC, Cook M, Larkin J, Marais R, Meneguzzi G, Sahai E, Marshall CJ (2011) Cancer Cell 20, 229-245 5. Fibroblast-led collective invasion of carcinoma cells with differing roles for RhoGTPases in leading and following cells Gaggioli C, Hooper S, Hidalgo-Carcedo C, Grosse R, Marshall JF, Harrington K, Sahai E (2007) Nat Cell Biol 9, 1392-1400 Awards 1999: Annual Congress of Dermatological Research (Magnaldo) 2007: European Society of Dermatological Research (Magnaldo) 2005: Prix jeune chercheur 2005 fondation Bettencourt-Schueller
-
Marie Hélène – Molecular mechanisms of neuronal plasticity in health and disease
MARIE Hélène

MARIE Hélène, CNRS Group Leader IPMC marie@ipmc.cnrs.fr 04 93 95 34 40 www.ipmc.cnrs.fr/?page=marie&lang=uk 
Molecular Mechanisms of Neuronal Plasticity in Health and Disease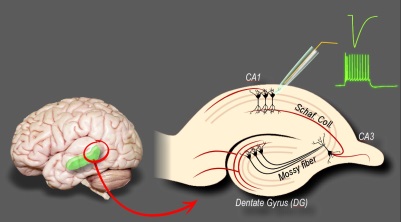 Electrophysiological recordings of hippocampal neurons from acute slices of Alzheimer’s disease mice to investigate neuronal dysfunction
Electrophysiological recordings of hippocampal neurons from acute slices of Alzheimer’s disease mice to investigate neuronal dysfunctionMemory is a fundamental brain function allowing an individual to adapt to his environment, to create his own and unique history. Identifying the cellular and molecular mechanisms that govern this process is one of the great challenges facing the neuroscientific community. Our team is working to elucidate these mechanisms and how they become defective in memory disorders such as in Alzheimer’s disease. Some of our recent work focused on the role of the transcription factor CREB in the molecular mechanisms of memory. We studied the implication of CREB by coupling behavioral, biochemical and electrophysiological analyses. We developed in vivo viral-mediated expression of specific proteins to evaluate their implication in memory processes. We also study transgenic mouse models of Alzheimer’s disease to characterize defects in synaptic plasticity of the hippocampus, a structure primarily affected in this disease. Using electrophysiology, we record hippocampal glutamatergic neurons to characterize in depth their functional deficits. We complete our functional analysis with a biochemical analysis of the hippocampus. We also evaluate the memory deficits of these models by behavioral studies. Finally, based on our results, we test new therapeutic strategies that are designed to prevent or delay this loss. Selected Publications
1. CREB is necessary for synaptic maintenance and learning-induced changes of the AMPA receptor GluA1 subunit. Middei S, Houeland G, Cavallucci V, Ammassari-Teule M, D’Amelio M, and Marie H. (2013) Hippocampus 23, 488-499 2. Computational modeling of the effects of amyloid-beta on release probability at hippocampal synapses. Romani A, Marchetti C, Bianchi D, Leinekugel X, Poirazi P, Migliore M, Marie H. (2013) Front. Comp. Neurosci. 7, 1-7 3. Hippocampal synaptic plasticity in Alzheimer’s disease: what have we learned so far from transgenic models? Marchetti C, Marie H. (2011) Rev Neurosci. 22, 373-402 4. Caspase-3 triggers early synaptic dysfunction in a mouse model of Alzheimer’s Disease. D’Amelio M, Cavallucci V., Middei S., Marchetti C., Pacioni S., Ferri A., Diamantini A., De Zio D., Carrara P., Battistini L., Moreno S., Bacci A., Ammassari-Teule M., Marie H., Cecconi F. (2011) Nat Neurosci. 14, 69-76 5. Chronic NGF deprivation results in mild deficits in hippocampal CA1, but severe deficits in dentate gyrus glutamatergic synaptic plasticity Houeland G., Romani A., Marchetti C., Amato G., Capsoni S., Cattaneo A., Marie H.(2010) J. Neurosci. 30, 13089-13094 Awards 2010: Young Investigator ATIP-AVENIR Award (France) 2009: New Investigator Alzheimer Association Grantee (USA) 2007: NARSAD Young Investigator Award (USA)
-
Martin Stéphane – Activity-dependent dynamics and roles of synaptic sumoylation
MARTIN Stéphane

MARTIN Stéphane, Inserm Group Leader IPMC martin@ipmc.cnrs.fr 0493953461 https://www.ipmc.cnrs.fr/cgi-bin/standard.cgi?descriptif=martin.txt&dossier1=equipes&dossier2=martin&site=inter&menu=1&ssmenu=16&lang=uk 
Activity-dependent dynamics and roles of synaptic sumoylation
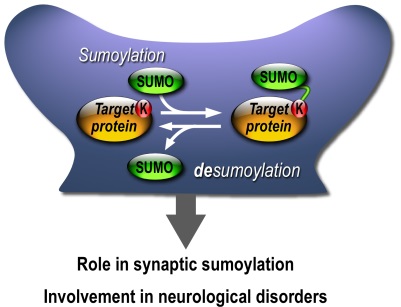 Investigating the function and dysfunction of synaptic sumoylation
Investigating the function and dysfunction of synaptic sumoylationSumoylation is a covalent and reversible enzymatic process that involves the conjugation of a small protein called SUMO to specific lysine residues of target proteins. Sumoylation was initially thought to target nuclear proteins where it is an essential regulator of their function. However, we discovered that multiple synaptic proteins are substrates for sumoylation (Martin et al., 2007, Nature). This raised the possibility that sumoylation, like other post-translational modifications (phosphorylation, ubiquitination…) play important roles in brain function. Intriguingly, among identified SUMO substrates are several proteins directly linked to neurological disorders.Our focus is now on synaptic sumoylation, a key posttranslational modification for many proteins including some involved in brain disorders. Using biochemical and state-of-the-art imaging techniques we are investigating the regulatory mechanisms of the SUMO pathway and functionally characterizing novel sumoylated proteins involved in intellectual disabilities.The wide range of state-of-the-art expertise available (proteomic, animal and imaging facilities) within the IPMC as well as the proximity of the Labex groups provide exceptional added values to our projects and we are confident that we will make real and meaningful advances in this competitive field of Neuroscience. Selected Publications
1. Activity-dependent regulation of the sumoylation machinery in rat hippocampal neurons. Loriol C, Khayachi A, Poupon G, Gwizdek C and Martin S. (2013) Biol Cell 105, 30-45 2. Developmental regulation and spatiotemporal redistribution of the sumoylation machinery in the rat Central Nervous System. Loriol C, Parisot J, Poupon G, Gwizdek C and Martin S. (2012) PLoS ONE 7, e33757 3. Emerging extranuclear roles for protein sumoylation in neuronal function and dysfunction. Martin S, Wilkinson K, Nishimune A and Henley JM. (2007) Nature Reviews Neuroscience 8, 948-959 4. Sumoylation Regulates Kainate Receptor Mediated Synaptic Transmission. Martin S, Nishimune A, Mellor J and Henley JM. (2007) Nature 447, 321-325 Awards 2012: ATIP+ CNRS 2009: Bettencourt-Schueller Foundation Prize 2008: ATIP CNRS
-
Nahon Jean-Louis – Genomics and Evolution in Neuroendocrinology (GENE)
NAHON Jean-Louis

NAHON Jean-Louis, CNRS Group Leader IPMC nahonjl@ipmc.cnrs.fr 04 93 95 77 53 http://www.ipmc.cnrs.fr 
Genomics and Evolution in Neuroendocrinology (GENE)
 Evolution, structure and function of the primate-specific genes. From gene sequencing (DNA helix) til functional characterization (biochip)
Evolution, structure and function of the primate-specific genes. From gene sequencing (DNA helix) til functional characterization (biochip)Diseases linked to energy intake-storage disequilibria are a major challenge for human health care. We are investigating the structural evolution, regulation and functions of genes involved in the central control of energy balance in mammals. Our favorite model concerns a peptide called “melanin-concentrating hormone” (MCH). Two relevant exemples of our research projects are presented below. 1. Central regulation of neuroendocrine functions Two MCH receptors (MCHR1 and MCHR2) have been found in humans whereas only MCHR1 has been identified in rodents. In order to study MCH signaling in an animal model mimicking the human situation we have generated a transgenic mouse model expressing human MCHR2 (KI-hMCHR2 mouse). We are currently analyzing the expression profile and phenotypes of homozygotic KI-hMCHR2 mice. 2. Genomics and Evolution By combining Molecular Biology, Phylogenetic and Bioinformatic techniques we have proposed an original scenario for the emergence in Hominoid lineage of two “chimaeric” genes, named PMCHL1 and PMCHL2. These “chimaeric” genes display differential expression in the human brain. One of our major objectives is to unravel their functional relevance in higher Primates. We are also investigating putative association between expression of the “primate-specific” genes and etiology of metabolic disorders and neurodegenerative diseases. Selected Publications
1. Melanin-concentrating hormone regulates beat frequency of ependymal cilia and ventricular volume. Conductier G*, Brau F*, Viola A*, Langlet F, Ramkuma N, Dehouck B, Lemaire T, Chapot R, Lucas L, Rovère C, Maitre P, Hosseiny S, Petit-Paitel A, Adamantidis A, Lakaye B, Risold PY, Prévot V**, Meste O**, Nahon JL**§, Guyon A**§ (2013) Nature Neurosci doi: 10.1038/nn.3401, 1-3 2. Variations in circulating inflammatory factors are related to changes in calorie and carbohydrate intakes early in the course of surgery-induced weight reduction. Dalmas E, Rouault C, Abdennour M, Rovere C, Rizkalla S, Bar-Hen A, Nahon JL, Bouillot JL, Guerre-Millo M, Clément K, Poitou C. (2011) Am J Clin Nutr. 94, 450-458 3. Genes regulated in MPTP-treated macaques and human Parkinson’s disease suggest a common signature in prefrontal cortex. Storvik M*, Arguel M.-J.*, Schmieder S., Delerue-Audegond A., Li Q., Qin C., Vital A., Bioulac B., Bross C.E., Wong G., Nahon JL **, Bezard E. ** (2010) Neurobiol. Dis. 38, 386-394 4. Glucose inhibition persists in hypothalamic neurons lacking tandem-pore K+ channels. Guyon A, Tardy MP, Rovère C, Nahon JL**, Barhanin J**, Lesage F. ** (2009) J Neurosci. 29, 2528-2533 5. Birth of two chimeric genes in the Hominidae lineage Courseaux A. and Nahon J.L (2001) Science (Human Genome Issue) 291, 1293-1297 Awards 2012: Prime d’Excellence Scientifique -CNRS (JLN) 2012: Danone Research Award (GC) 2010: Société Française de Nutrition (GC) 2009: Prix de la Société de Neuroendocrinologie (GC)
-
Noselli Stéphane – Epithelial morphogenesis and left-right asymmetry in Drosophila
NOSELLI Stéphane

NOSELLI Stéphane, CNRS Group Leader iBV noselli@unice.fr04 92 07 64 33 http://ibv.unice.fr/EN/equipe/noselli.php 
Epithelial morphogenesis and left-right asymmetry in Drosophila
 Epithelial morphogenesis, cell migration and left-right asymmetry in Drosophila
Epithelial morphogenesis, cell migration and left-right asymmetry in DrosophilaUsing Drosophila, our team made several original contributions leading to the emergence of new fields (Dorsal closure, JNK, Jak/Stat, novel roles of ECM, L/R asymmetry). The diversity of biological questions studied provides an exciting scientific environment, with more than 40 students and post-docs trained.JNK, Dorsal Closure (DC), reprogramming: our team identified the physiological role of the conserved JNK pathway, controlling epithelial morphogenesis during DC and cell reprogramming to release tissue tension. Current work characterizes novel JNK target genes and the mechanisms of cell reprogramming.Extracellular matrix, Jak/Stat, cell invasion: we co-discovered the Drosophila Jak/Stat receptor and its role in cell migration during oogenesis. Our current results reveal a novel and complex assembly of basement membrane BM, redefining its role in tissue homeostasis and signaling. We also reveal a novel role of specific blood cells in matrix formation important for stem cell niche control.Left-Right asymmetry: we established Drosophila as a L/R model, through the identification of the conserved Myosin ID gene (MyoID). Recent work showed a coupling between L/R patterning and apoptosis, and identified HOX/Abd-B as a ‘master’ gene controlling L/R asymmetry. Our current work studies the coupling of L/R and PCP patterning as well as characterizing novel L/R asymmetry genes. Selected Publications
1. Drosophila Left/Right asymmetry establishment is controlled by the Hox gene Abdominal-B Coutelis JB, Géminard C, Spéder P, Suzanne M, Petzoldt A and Noselli S. (2013) Dev Cell 24, 89-97 2. Coupling of apoptosis and L/R patterning controls stepwise organ looping Suzanne, M., Petzoldt, A.G., Spéder, P., Coutelis, J.B., Steller, H. and Noselli, S (2010) Curr Biol 20, 1173-1178 3. JNK signalling controls remodelling of the segment boundary through cell reprogramming during Drosophila morphogenesis Gettings M, Serman F, Rousset R, Bagnerini P, Almeida L, Noselli S. (2010) PLoS Biol 8, 1000390 4. Type ID unconventional myosin controls left-right asymmetry in Drosophila Spéder*, P., Adam*, G. & Noselli, S (2006) Nature 440, 803-807 5. hemipterous encodes a novel Drosophila MAP kinase kinase, required for epithelial cell sheet movement Glise, B., Bourbon, H., and Noselli, S (1995) Cell 83, 451-461 Awards 2013: Grand Prix Mottart, Académie des Sciences 2008: CNRS Silver Medal 2011: EMBO Young Investigator Program (EMBO YIP) 1999: ATIP CNRS, Developmental Biology 1998: CNRS Bronze Medal
-
Panabières Franck – Plant oomycete interactions
PANABIERES Franck

PANABIERES Franck, INRA Group Leader ISA franck.panabieres@sophia.inra.fr04 92 38 65 18 http://www6.paca.inra.fr/institut-sophia-agrobiotech_eng/Research-teams/IPO 
Plant oomycete interactions
 Research is conducted on both plant and pathogen, using a multi-faceted approach from cell biology to functional genomics and biochemistry
Research is conducted on both plant and pathogen, using a multi-faceted approach from cell biology to functional genomics and biochemistryOomycetes are eukaryotic microorganisms that include severe plant pathogens, and then constitute a major threat to agriculture and environment worldwide.Despite similar physiological and ecological traits shared with fungi, oomycetes display phylogenetic affinities with diatoms and brown algae among Chromalveolates, that include dinoflagellates and several animal pathogens (Plasmodium,Toxoplasma,Theileria…). As a consequence of this taxonomical position, most traditional fungicide molecules are generally poorly efficient, because they have been developed against true pathogenic fungi. In addition, adaptive potential of oomycetes in response to their environment leads to a rapid breakdown of varietal resistances. So, the development of integrated management strategies that would be more efficient and environmentally friendly implies a better knowledge on the mechanisms that govern oomycete physiology and pathogenicity.In this context, our team constructs multidisciplinary strategies to decipher the molecular dialog occurring between the oomycete pathogen and its plant host. We develop functional genomics approaches that rely on complete oomycete and plant genomes. They allow defining the interaction transcriptome from both partners and identifying key regulators, as well as identifying molecules that stimulate plant innate defense mechanisms. Selected Publications
1. The Phytophthora parasitica RXLR effector Penetration-Specific Effector 1 favours Arabidopsis thaliana infection by interfering with auxin physiology. Evangelisti E, Govetto B, Minet-Kebdani N, Kuhn ML, Attard A, Ponchet M, Panabières F, Gourgues M (2013) New Phytol Apr 17. doi: 10.1111/nph.12270. , [Epub ahead of print]. 2. Ecosystem screening approach for pathogen-associated microorganisms affecting host disease Galiana E, Mura C, Marais A. Industri B, Arbiol G, Ponchet M (2011) App Environ Microbiol 77, 6069-6075 3. An Arabidopsis (malectin-like) leucine-rich repeat receptor-like kinase contributes to downy mildew disease. Hok S, Danchin EG, Allasia V, Panabières F, Attard A, Keller H (2011) Plant Cell Environ 34, 1944-1957 4. The immediate activation of defense responses in Arabidopsis roots is not sufficient to prevent Phytophthora parasitica infection. Attard A, Gourgues M, Callemeyn-Torre N, Keller H (2010) New Phytol 187, 449-460 5. Cellular and molecular characterization of Phytophthora parasitica appressorium mediated penetration. Kebdani N, Pieuchot L, Deleury E, Panabières F, Le Berre JY, Gourgues M (2010) New Phytol 185, 248-257
-
Poirié Marylène – Evolution and Specificity of Multitrophic Interactions (ESIM)
POIRIE Marylène

POIRIE Marylène, UNS Group Leader ISA marylene.poirie@sophia.inra.fr0492386409 http://www6.paca.inra.fr/institut-sophia-agrobiotech/Equipes-de-recherche/ESIM 
Evolution and Specificity of Multitrophic Interactions (ESIM)
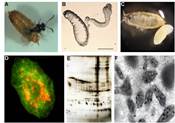 L. boulardi wasp: emerging adult (A) larvae (C) venom gland (B). RhoGAP toxin in host hemocytes (D) on venom vesicles (F). 2D of venom (E).
L. boulardi wasp: emerging adult (A) larvae (C) venom gland (B). RhoGAP toxin in host hemocytes (D) on venom vesicles (F). 2D of venom (E).The team mainly focuses on the immune interactions between insects hosts (Drosophila species) and their parasitoid wasps (Hymenoptera) as a model of immune suppressive eukaryotic parasites. We aim to characterize at the molecular and cellular level both the molecules/mechanisms involved in parasitoid virulence and those responsible for host resistance, as well as to identify the bases of the specificity of the interaction. Resistance may be linked with signaling networks involved in hemocyte proliferation/differentiation. Parasitoid virulence is achieved through injection of venom components, notably extra-cellular RhoGAPs toxins that are internalized inside host hemocytes and target Rac proteins. The transport of the RhoGAPs inside hemocytes through recently identified venom vesicles (venosomes) and the nature of the involved receptors is one of our main subject area. Evolutionary aspects of these interactions lie also at the heart of the research undertaken.We also recently developed research on the immune components in a new insect model, the pea aphid. Thanks to its clonal reproduction and long-lasting symbioses with bacteria, this model allows us to finely address the question of how symbionts can shape the immunity of their host, in the context of the link between immunity and metabolism. Selected Publications
1. Tracing back the nascence of a new sex determination pathway to the ancestor of bees and ants. Schmieder S, Colinet C and Poirié M. (2012) Nat. Commun. 3, 895 2. Extracellular superoxide dismutase in insects: characterization, function and inter-specific variation in parasitoid wasps’ venom. Colinet D, Cazes D, Belghazi M, Gatti JL, Poirié M. (2011) J Biol Chem 286(46), 40110-21 3. Colinet D., Schmitz A., Cazes D., Gatti JL, Poirié M. The origin of intraspecific variation of virulence in an eukaryotic immune suppressive parasite. Colinet D, Schmitz A, Cazes D, Gatti JL, Poirié M. (2010) PLoS Pathog 6(11) , e1001206. 4. Variation of Leptopilina boulardi success in Drosophila hosts: what is inside the black box? Dubuffet A, Colinet D, Anselme C, Dupas S, Carton Y and Poirié M. (2009) Adv Parasitol 70, 148-188 5. Convergent use of RhoGAP toxins by eukaryotic parasites and bacterial pathogens. Colinet D, Schmitz A, Depoix D, Crochard D and Poirié M. (2007) PloS Pathog 3(12), e203 Awards 2009: Prize of the French National Academy of Sciences in Integrative Sciences – Balachowski prize of Entomology 2009-2011: “Jeune Equipe INRA “- thème “Immune interactions between insect hosts and insect parasitoids : molecular, cellular and evolutionary aspects”
-
Rassoulzadegan Minoo – RNA-mediated epigenetic heredity
RASSOULZADEGAN Minoo

RASSOULZADEGAN Minoo, CNRS Group Leader iBV minoo@unice.fr0492076412 http://ibv.unice.fr 
RNA-mediated epigenetic heredity
 10 days old, Kit mutants and paramutants progenies (back-ground n Agouti mouse strain).
10 days old, Kit mutants and paramutants progenies (back-ground n Agouti mouse strain).Our laboratory established the first mouse models of an epigenetic heredity distinct from the Mendelian rules. Small noncoding (snc) RNA molecules with sequence homology to the transcript were shown to act as transgenerational signals leading to the establishment of the modified phenotypes. Several of the observed variants reproduce human pathologies with a clear familial distribution but without a Mendelian determinant identified, such as heart hypertrophy and a form of diabetes. The modified phenotypes independently observed for three loci, Kit, Sox9 and Cdk9 result from a transcriptional modulation of the locus activity. Our current work aims at the establishment of the molecular mechanisms of the variation using two types of models, the early embryo in which paramutation is triggered by microinjection of the signalling RNA and identification of the vectors in transmission of the acquired characters, in which we have demonstrated a response to the same sncRNA identical to that observed in paramutation. The analysis focuses on the following parts of the signalling cascade: a requirement for cytosine methylation in the inducing sncRNAs by the RNA methyltransferase Dnmt2. We are also exploring the possibility of RNA-signalling and transgenerational maintenance of other phenotypes including comportmental variations for which evidence of paternal inheritance has been established. Selected Publications
1. RNA-Mediated Epigenetic Heredity Requires the Cytosine Methyltransferase Dnmt2. Kiani J, Grandjean V, Liebers R, Tuorto F, Ghanbarian H, Lyko F, Cuzin F, Rassoulzadegan M. (2013 ) PLoS Genet. May;9(5): e1003498. 2. Novel small noncoding RNAs in mouse spermatozoa, zygotes and early embryos. Kawano M, Kawaji H, Grandjean V, Kiani J, Rassoulzadegan M. (2012) PLoS One. ;7(9), e44542. 3. The miR-124-Sox9 paramutation: RNA-mediated epigenetic control of embryonic and adult growth. Grandjean V, Gounon P, Wagner N, Martin L, Wagner KD, Bernex F, Cuzin F, Rassoulzadegan M. (2009) Development. Nov;136(21), 3647-55. 4. RNA induction and inheritance of epigenetic cardiac hypertrophy in the mouse. Wagner KD, Wagner N, Ghanbarian H, Grandjean V, Gounon P, Cuzin F, Rassoulzadegan M. (2008) Dev Cell. Jun;14(6), 962-9. 5. RNA-mediated non-mendelian inheritance of an epigenetic change in the mouse. Rassoulzadegan M, Grandjean V, Gounon P, Vincent S, Gillot I, Cuzin F. (2006 ) Nature. May 25;441(7092): 469-74. Awards 2011: Prix Doyen Lepine de la ville de Nice 2009: Elected EMBO Member 1987: Prix FRM (Biologie Moléculaire) 1985: Prix Lacassagne
-
Ricci Jean Ehrland – Metabolic control of cell deaths
RICCI Jean-Ehrland

RICCI Jean-Ehrland, Inserm Group Leader C3M ricci@unice.fr04 89 06 43 04 http://www.unice.fr/c3m/EN/Equipe3.html 
Metabolic control of cell deaths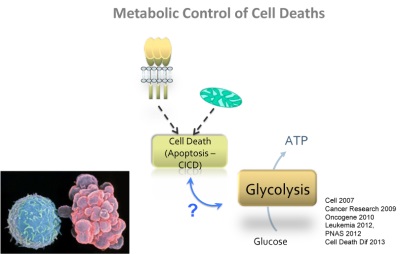 We are investigating how targeting glucose metabolism can enhance cell death (apoptotic and a non-apoptotic forms of death called CICD)
We are investigating how targeting glucose metabolism can enhance cell death (apoptotic and a non-apoptotic forms of death called CICD)The main drawback of chemotherapies is their lack of specificity toward cancer cells. In addition cancer cells are very diverse. However they are sharing a few commune features including escape from cell death and a particular metabolism (so-called the Warburg effect). In our team we aim to understand how the modulation of cancer cell metabolism could improve therapies (increase cell death) and modulate anticancer immune response. We demonstrated that the inhibition of glucose metabolism can not only enhances the chemotherapeutic toxicity but it can also allow the immune system to react against the tumor (leading to an anti-cancer immune response that participates in a long term protection of the patient against the tumor). In collaboration with chemists, we are now developing original glycolytic inhibitors to enhance this effect and increase its specificity. In parallel we observed that out of the 10 glycolytic enzymes needed for glucose metabolism, one (GAPDH) is very particular as it can help the cell to escape non apoptotic from of death. We are deciphering its mechanisms of action. Our research is conducted in cell lines, pre-clinical models as with patient samples (in close relation with the clinical service of Nice and Monaco hospitals). Selected Publications
1. GAPDH binds active Akt leading to Bcl-xL increase and escape from caspase-independent cell death. 1. Jacquin M.A., Chiche J., Zunino B., Bénéteau M., Meynet O., Pradelli L.A. Marchetti S., Cornille A., Carles M. and J-E Ricci. (2013) Cell Death Differ in press, in press 2. The combination of glycoysis inhibition with chemotherapy results in an antitumor immune response. 2. Bénéteau M., Zunino B., Jacquin M.A., Chiche J., Meynet O., Pradelli L.A., Marchetti S., Cornille A., Carles M. and J-E Ricci. (2012) Proc Natl Acad Sci U S A. 109, 20071-6 3. Cancer metabolism: current perspectives and future directions. 4. Munoz-Pinedo C, El Mjiyad N and J-E Ricci (2012) Cell Death and Disease 3, e248 4. Glycolysis inhibition targets Mcl-1 to restore sensitivity of lymphoma cells to ABT-737-induced apoptosis. 3. Meynet O., Bénéteau M., Jacquin M.A., Pradelli L.A., Cornille A., Carles M. and J-E Ricci. (2012) Leukemia 26, 1145-7 5. Glycolysis inhibition sensitizes tumor cells to death receptors-induced apoptosis by AMP kinase activation leading to Mcl-1 block in translation. 10. Pradelli, L.A., M. Beneteau, C. Chauvin, M.A. Jacquin, S. Marchetti, C. Munoz-Pinedo, P. Auberger, M. Pende, and J.E. Ricci. (2010) Oncogene 29, 1641-52 Awards 2012: Award “Arloing, Courmont” from Pasteur Institute in Lyon, France; 2010: Award from the 7th EU Workshop on Cell Death; 2006: Awarded of the INSERM/AVENIR program
-
Robichon Alain – Genome Plasticity and Environment
ROBICHON Alain

ROBICHON Alain, CNRS Group Leader ISA alain.robichon@sophia.inra.fr 04 92 38 64 19 http://www6.paca.inra.fr/institut-sophia-agrobiotech 
Genome Plasticity and Environment
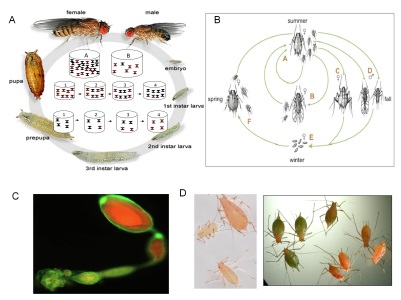 Epigenetic variation in drosophila m.(A) and aphid A pisum (B-D)
Epigenetic variation in drosophila m.(A) and aphid A pisum (B-D)Up to recently, the dogma postulates that evolution and genome plasticity are written in the genetic code. In contrast, many recent reports describe that identical genomes might lead to different heritable characters depending on environmental factors, thus opposing genetic determinism and epigenetic response. Surprisingly and of considerable interest, we found that the A. pisum that alternates clonality and sexuality, generates a repertoire of phenotypic variants. We recently found solid evidence for a role of methylation of the aphid genome in the regulation of environmentally induced phenotypes. We are interested in investigating the potential of stable epigenetic marks that might contribute to lasting and heritable phenotypes. Our objective is to determine whether patterns of DNA methylation might be heritable across clonality and also whether this heritability might pass the sexual barrier to be transmitted in the newly emerged clonal generations the next spring. Our team is particularly interested to investigate the mechanisms of the epigenetic regulation of adaptive traits of insects in fluctuating environment. Using genetic and biochemical tools the aim of our project consists in analyzing the covalent modifications of DNA, their functional roles and their heritable transmission that confers the passage of an acquired phenotype to the next generations in clonality context. Selected Publications
1. Profiling the repertoire of phenotypes influenced by environmental cues that occur during asexual reproduction Dombrovsky A., Arthaud L., Ledger T.N., Tares S. and Robichon A.. (2009) Genome Res 19(11), 2052-63. 2. Genome Sequence of the Pea Aphid Acyrthosiphon pisum The International Aphid Genomics Consortium (2010) PLoS Biol. 8(2), e1000313. 3. Light- induced electron transfer and ATP synthesis in a carotene synthesizing insect Valmalette J.C., Dombrovsky A., Brat P., Christian Mertz C., Capovilla M., Alain Robichon A. (2012) Scientific Reports 2, article number 579/ doi : 10.1038/srep00579 4. Analysis of carotenoid compounds in aphids by Raman imaging and mass spectrometry. Bratt, P., Valmalette, J. C., Mertz, C., de Sousa, G., Dombrovsky, A., Capovilla, M., and Robichon, A. (2012) Nature Protocol Exchange none, doi:10.1038/protex.2012.047 5. Photosynthesis-like process found in insects Lougheed K. (2012) Nature News 11214, doi:10.1038/nature.2012.11214
-
Schedl Andreas – Molecular programs controlling development and tissue homeostasis
SCHEDL Andreas

SCHEDL Andreas, Inserm Group Leader iBV schedl@unice.fr 0492076401 http://ibv.unice.fr/EN/equipe/schedl.php 
Molecular programs controlling development and tissue homeostasis
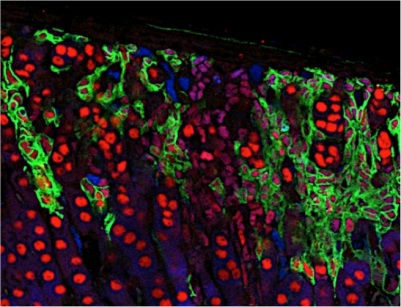
Lineage tracing analysis identifies a novel stem/progenitor cell population in adult mice.
Tissue repair in most organs is guaranteed through resident stem/progenitor cells. Stem cells are essential for organ renewal and lack of activation or depletion of the stem cell pool can lead to progressive organ failure. In contrast overactivation of stem cells and their transiently proliferating derivatives can cause overgrowth and cancer. Understanding stem cell biology is therefore of very high importance from both a biological and a medical point of view.Development and tissue maintenance are highly interrelated processes and molecular programs driving development and differentiation are also triggered when stem cells become activated to replace damaged or lost cells. In our research program we try to understand the transcriptional control underlying tissue development, define stem/progenitor cells in the adult organism and determine the signaling pathways involved in their maintenance and activation. As a model of choice we use the mouse, as its physiology largely reflects that of the human body and as it allows easy manipulation of its genome using transgenic and gene-targeting techniques. Selected Publications
1. SOX9 Controls Epithelial Branching by Activating RET Effector Genes during Kidney Development Reginensi A, Clarkson M, NeirijnckY, Lu B, Ohyama T, Groves AK, Sock E, Wegner M, Costantini F, Chaboissie MC, Schedl A (2011) Hum. Mol. Genet. 20, 1143-1153 2. Renal abnormalities and their developmental origin. Schedl A (2007) Nature Rev. Genet.8, 791-802 3. Coronary vessel development requires activation of the TrkB neurotrophin receptor by the Wilms’ tumor transcription factor Wt1 Wagner N., Wagner KD., Theres H., Englert C., Schedl A*. and Scholz H (2005) Geners & Dev 19, 2631-2642 4. Sox9 induces testis development in XX transgenic mice Vidal VP, Chaboissier MC, de Rooij DG, Schedl A (2001) Nat. Genet. 28, 216-217 5. Two alternatively spliced isoforms of WT1 have distinct functions during sex determination and nephron formation. Hammes A, Guo J, Lutsch G, Landrock D, Ziegler A, Gubler MC, Schedl A (2001) Cell 106, 319-329 Awards 2009: Award of the French kidney foundation (Fondation du Rein) 2003: Avenir (INSERM, France 2002: Philippe Leverhulme Prize (UK) 2001: EMBO Young Investigator Programme
-
Studer Michèle – Genetics of mouse cortical development
STUDER Michèle

STUDER Michèle, Inserm Group Leader iBV michele.studer@unice.fr0492 07 6419 http://ibv.unice.fr/EN/equipe/studer.php 
Genetics of mouse cortical development
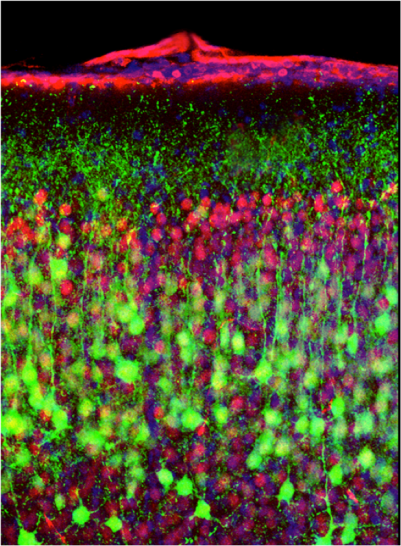 In utero electroproration of EGFP at E14.5 and taken at P8 showing the soma and dendrites of upper layer neurons of the mouse cortex.
In utero electroproration of EGFP at E14.5 and taken at P8 showing the soma and dendrites of upper layer neurons of the mouse cortex.The major aim of our research is to elucidate the key mechanisms of neurogenesis in the developing and post-natal mouse cerebral cortex. During corticogenesis, the remarkable array of different neuronal cell types is generated from a population of multipotent stem and progenitor cells following a precise spatial and temporal pattern, before being assembled in areal- and laminar-specific maps and circuits. We are interested in understanding the cellular and molecular mechanisms by which these different neuronal cell types are temporally and spatially regulated through the action of defined regionalized and coordinated differentiation programs.We found that a family of nuclear receptors is required in balancing the neocortex into motor and sensory areas, in controlling the generation and specification of corticospinal motor neurons, in axonal outgrowth, in neuronal migration, and in cortical interneuron migration and cell-type specification.By combining several in vivo and in vitro molecular and cellular approaches, including mouse genetics, in utero elctroporation, biochemical assays, neural stem cell cultures, but also morphological and behavioural analyses, we aim to contribute in dissecting signalling pathways and molecular cascades involved in cortical neuronal cell-type specification and establishment of functional cortical circuits during mouse brain development. Selected Publications
1. The nuclear receptors COUP-TF: a long lasting experience in forebrain assembly. Alfano C., Kawssar H., Magrinelli E. and Studer M. (2013) Cell Mol Life Sci.Mar 23., [Epub ahead of print] 2. COUP-TFI promotes radial migration and proper morphology of callosal neurons by repressing Rnd2 expression. 6. Alfano C., Viola L., Heng J.I.T., Pirozzi M., Clarkson M., Flore G., De Maio A., Schedl A., Guillemot F. and Studer M. (2011) Development 138, 4685-4697 3. Loss of COUP-TFI alters the balance between caudal ganglionic eminence- and medial ganglionic eminence-derived interneurons and results in resistance to epilepsy. Lodato S., Tomassy Srubek G., De Leonibus E., Uzcategui Y.G., Andolfi G., Armentano M., Touzot A., Gaztelu J. M., Arlotta P., Menendez de la Prida L. and Studer M. (2011) J Neurosci. 31(12), 4650-4662 4. Area-specific temporal control of corticospinal motor neuron differentiation by COUP-TFI. Tomassy Srubek G., De Leonibus E., Jabaudon D., Lodato S., Alfano C., Mele A., Macklis J.D. and Studer M. (2010) PNAS 107(8), 3576-3581 5. COUP-TFI regulates the balance of cortical patterning between frontal/motor and sensory areas. Armentano M., Chou S. J., Srubek Tomassy G., Leingärtner A., O’Leary D.D.M. and Studer M. (2007) Nat Neurosci.10, 1277-1286 Awards 2011: Equipe labélisée FRM 2009: ANR Senior Chaire d’Excellence 1997: MRC (Medical Research Council) Career Development Award
-
Tanti Jean-François/Cormont Mireille – Cellular and Molecular Pathophysiology of Obesity and Diabetes
TANTI Jean-François/CORMONT Mireille

TANTI Jean-François, Inserm Group Leader C3M tanti@unice.fr0489064237 http://www.unice.fr/c3m/EN/Equipe7.html 
CORMONT Mireille, Inserm Group Leader C3M cormont@unice.fr 0489064234 http://www.unice.fr/c3m/EN/Equipe7.html 
Cellular and Molecular Physiopathology of Obesity and Diabetes
 Our team by using cells and animal models deciphers the mechanisms linking adipose tissue dysfunction to insulin resistance and tumor growth
Our team by using cells and animal models deciphers the mechanisms linking adipose tissue dysfunction to insulin resistance and tumor growthInflammatory and hypoxic stresses as well as senescence develop in AT during obesity. Activation of stress-sensing pathways in obese AT alters insulin signaling and the trafficking of the glucose transporter Glut4 decreasing the metabolic functions of adipocytes leading to insulin resistance and type 2 diabetes development. Further adipose tissue stresses maybe involved in obesity–related carcinogenesis. Our team by using cells and animal models deciphers the dysfunction of adipose tissue metabolism linked to obesity and diabetes. Our objectives are: 1. To identify the cellular and molecular mechanisms involved in insulin resistance induced by AT inflammatory and hypoxic stresses. We study: • The implication of RabGTPase in Glut4 trafficking and in the regulation of the gluco-lipidic metabolism. • The implication of microRNAs in the control of the metabolic functions of adipocytes and their deregulations in obesity. • The implication of stress kinases in adipose tissue inflammation and insulin resistance. • The implication of hypoxia in adipocyte insulin resistance. 2. To understand the pathophysiological mechanisms responsible for the association between obesity and cancer. We study: • The effect of insulin sensitizing drugs on tumor development Our goal is to identify new therapeutic targets for the treatment of obesity-associated pathologies. Selected Publications
1. Regulated in development and DNA damage responses -1 (REDD1) protein contributes to insulin signaling pathway in adipocytes. Regazzetti C, Dumas K, Le Marchand-Brustel Y, Peraldi P, Tanti JF, Giorgetti-Peraldi S. (2012) PLoS One. 12, e52154 2. Metformin, independent of AMPK, induces mTOR inhibition and cell-cycle arrest through REDD1. Ben Sahra I, Regazzetti C, Robert G, Laurent K, Le Marchand-Brustel Y, Auberger P, Tanti JF, Giorgetti-Peraldi S, Bost F. (2011) Cancer Res 71, 4366-4372 3. Deficiency in the extracellular signal-regulated kinase 1 (ERK1) protects leptin-deficient mice from insulin resistance without affecting obesity. Jager J, Corcelle V, Grémeaux T, Laurent K, Waget A, Pagès G, Binétruy B, Le Marchand-Brustel Y, Burcelin R, Bost F, Tanti JF. (2011) Diabetologia 54, 180-189 4. Tpl2 kinase is upregulated in adipose tissue in obesity and may mediate interleukin-1beta and tumor necrosis factor-{alpha} effects on extracellular signal-regulated kinase activation and lipolysis. Jager J, Grémeaux T, Gonzalez T, Bonnafous S, Debard C, Laville M, Vidal H, Tran A, Gual P, Le Marchand-Brustel Y, Cormont M, Tanti JF. (2010) Diabetes 59, 61-70 5. Involvement of TNF-alpha in abnormal adipocyte and muscle sortilin expression in obese mice and humans. Kaddai V, Jager J, Gonzalez T, Najem-Lendom R, Bonnafous S, Tran A, Le Marchand-Brustel Y, Gual P, Tanti JF, Cormont M. (2009) Diabetologia 52, 932-940 -
Tartare-Deckert Sophie – Microenvironment, signaling and Cancer
TARTARE-DECKERT Sophie

TARTARE-DECKERT Sophie, Inserm Group Leader C3M tartare@unice.fr 04 89 06 43 10 http://www.unice.fr/c3m/EN/Equipe11.html 
Microenvironment, Signaling and Cancer Research workflow of team “MicroCan”
Research workflow of team “MicroCan”Our laboratory is interested in the crosstalk between cancer cells and stroma within the lymph node microenvironment in two models, melanoma, an aggressive form of skin cancers and lymphoma. The lymph node is the first locus of expansion for invasive melanoma and a pipeline for distant metastasis as well as one niche of malignant B-cells. During these past years, our team achieved important discoveries such as the identification of secreted factors from the microenvironment that promote Epithelial to Mesenchymal Transition (EMT) and inactivate p53 in melanoma. The team also contributed to characterization of survival pathways that operate in normal and pathological leukocytes. Ongoing projects are pursuing our observations using relevant pre-clinical models and aim at deciphering how lymphatic stromal cells impact on tumor survival. We are performing candidate gene and unbiased screen approaches for new microenvironment mediators of therapeutic response. In addition, we are exploring pathways operating within melanomas that enable them to invade lymphatics and to execute various steps of metastasis. Our work will bring a more comprehensive view of tumor-stroma communications and better understanding of melanoma and lymphoma malignancies. Our findings might have potential clinical implications for identification of novel biomarkers, potential targets and management of diseases. Selected Publications
1. A novel FRET flow cytometry method for detection of actin dynamics on resting and activated T cell Larbret F, Dubois N, Brau F, Guillemot E, Mahiddime K, Tartare-Deckert S, Verhasselt V and Deckert M. (2013) J Leuk Biol In press 2. The epithelial-mesenchymal transition (EMT) regulatory factor Slug (SNAI2) is a downstream target of SPARC and AKT in promoting melanoma cell invasion Fenouille N, Tichet M, Dufies M, Pottier A, Mogha A, Soo J K, Rocchi S, Malavialle A, Khammari A, Lacour J-P, Galibert MD, Ballotti R, Deckert M and Tartare-Deckert S. (2012) PLoS One 7, e40378 3. SPARC functions as an anti-stress factor by inactivating p53 through Akt-mediated MDM2 phosphorylation to promote melanoma cell survival Fenouille N, Puissant A, Tichet M, Zimniak G, Abbe P, Mallavialle A, Rocchi, S., Ortonne JP, Deckert M, Ballotti R, and Tartare-Deckert S. (2011) Oncogene 30, 4887-900 4. Permanent activation of the Fyn/Erk axis mediates Imatinib resistance in Chronic Myelogenous Leukemia cells through increased expression of intracellular SPARC Fenouille N, Puissant A, Dufies M, Robert G, Jacquel A, Ohanna M, Deckert M, Pasquet JM, Mahon FX, Cassuto JP, Raynaud S, Tartare-Deckert* S and Auberger* P. (2010) Cancer Res 70, 9659-9670 5. Spleen tyrosine kinase functions as a tumor suppressor in melanoma cells by inducing senescence-like growth arrest Bailet O, Fenouille N, Abbe P, Robert G, Rocchi S, Gonthier N, Denoyelle C, Ticchioni, M. Ortonne, JP, Ballotti, R, Deckert M and Tartare-Deckert S. (2009) Cancer Res 69, 2748-2756
-
Thérond Pascal – Secretion and Signaling of Morphogens in Drosophila development
THEROND Pascal

THEROND PASCAL, CNRS Group Leader iBV therond@unice.fr 0630946639 http://ibv.unice.fr/EN/equipe/therond.php 
Secretion and Signaling of Morphogens in Drosophila development
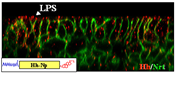 Confocal Z section showing Hedgehog (in red) aggregates (LPS) enriched at the apical side of embryonic epithelial cells (baso-lateral membrane)
Confocal Z section showing Hedgehog (in red) aggregates (LPS) enriched at the apical side of embryonic epithelial cells (baso-lateral membrane)We focus our studies on two molecules, the secreted cholesterol-modified Hedgehog (Hh) and Wingless (Wg) proteins, key molecules that control the pattern formation of multicellular organisms.We use genetic, biochemical and dynamic imaging approaches in Drosophila to identify and characterize molecules that modulate the cellular responses to Hh and Wg. We identified a protein module which governs proteolysis and access to the nucleus of the Ci/Gli transcription factor that mediates Hh signal (Robbins et al., Cell 1997; Ruel et al., NCB 2003). Moreover, our work highlighted the importance of the proteoglycans, of the extra-cellular matrix, in the internalization of the ligand-receptor complex for Hh signaling and Wg trafficking (Gallet et al., Dev. Cell 2008). Recently we developed new dynamic imaging tools to understand how the extra-cellular gradient of Hh is converted into an intra-cellular gradient of Ci/Gli activity (Ranieri et al., Dev. Cell 2012 and unpublished data).Our current research interests also include the spreading of Hh in the epitheliums (Briscoe and Thérond, Nat Rev Mol Cell Biol, 2013). We showed that the cholesterol adduct on Hh is necessary for its controlled planar spreading. We showed that the spread of Hh into the extracellular space is contingent upon its assembly in large soluble multimers and glypican activity (Gallet et al., Dev. Cell, 2003). We pioneered the concept that overall morphogen gradient is a cellular summation of pools secreted by different routes (apical and basolateral) and favor the existence of several carriers for Hh transportation (Ayers et al., Dev. Cell, 2010). We are currently isolating these different carriers and identifying the cellular machinery involved in Hh and Wg secretion. Selected Publications
1. The mechanisms of Hedgehog signaling and its roles in development and disease. Briscoe J. and Thérond P.(2013) Review in Nat Rev Mol Cell Biol. 14, 1-12 2. Distinct Phosphorylations on Kinesin Costal-2 Mediate Differential Hedgehog Signaling Strength. Ranieri, N., Ruel, L., Gallet, A., Raisin S., and Thérond P. (2012) Developmental Cell 22, 279-294 3. The long range Hedgehog Gradient Is Formed In the Apical Extracellular Space by the Glypican Dally and the Hydrolase Notum. Ayers K., Gallet A., Staccini-Lavenant L., and Thérond, P. (2010) Developmental Cell 18, 605–620 4. The Cellular Trafficking of the Glypican Dally-like Is Required for Full-Strength Hedgehog Signaling and Wingless Transcytosis. Gallet, A.,Staccini-Lavenant L. and Thérond P.(2008) Developmental Cell 14, 1-14 5. Phosphorylation of the atypical kinesin Costal2 by the kinase Fused induces the partial disassembly of the Smoothened-Fused-Costal2-Cubitus interruptus complex in Hedgehog signalling Ruel L., Gallet A., Raisin S., Truchi A., Lavenant L., Cervantes A. and Thérond P. (2007) Development 134, 3677-3689 Awards 2009: Victor Noury Prize from the French Academy of Science 2004: Marie Curie Host for Research Training 2001: EMBO Young Investigator Program award 1998: Recipient of the ATIPE Programme in Cell Biology from CNRS
-
Trabucchi Michele – Control of Gene Expression
TRABUCCHI Michele

TRABUCCHIMichele, Inserm Group Leader C3M michele.trabucchi@unice.fr04 89 064260 http://www.unice.fr/c3m/EN/Equipe10.html 
Control of gene expression
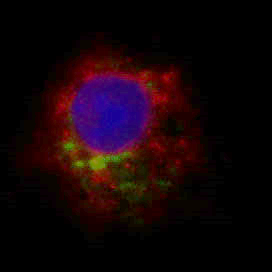 FISH detecting a small RNA (in green) expression in GW182-positive P-bodies (in red) of macrophage foam cells
FISH detecting a small RNA (in green) expression in GW182-positive P-bodies (in red) of macrophage foam cellsUncontrolled and chronic inflammation is nowadays considered a major component of many widely occurring diseases, including asthma, atherosclerosis and peripheral vascular disease, Alzheimer’s disease, type 2 diabetes, and cancer. Innate immune and inflammatory response significantly contributes to onset, progression, tissue dysfunction, and ultimately organ failure in these diseases. The study of the mechanisms regulating the amplitude and the consecutive resolution of the inflammation may provide new insight into the pathogenesis of inflammatory diseases allowing the exploration of novel targets for the treatment and the prevention of many disorders.We are investigating the molecular strategies used to generate genome-wide, integrated gene expression responses to the vast signaling network that regulates inflammatory response in macrophages. To achieve our goal we are using genetic, biochemical and biological approaches to define the gene expression strategies used by the human body in specific pathological contexts. Our work has revealed unexpected gene-specific strategies that link regulated gene responses to other cellular response programs, including cell signaling. Defining these strategies has suggested new approaches to diseases, including diabetes, arteriosclerosis, and several prevalent forms of cancer. Selected Publications
1. Let-7b/c enhance the stability of a tissue-specific mRNA during mammalian organogenesis as part of a feedback loop involving KSRP. E. Repetto, P. Briata, N. Kuziner, B.D. Harfe, M.T. McManus, R. Gherzi, M.G. Rosenfeld, M. Trabucchi (2012) Plos Genetics 8, e1002823 2. DICER- and AGO3-dependent generation of retinoic acid-induced DR2 Alu RNAs regulates human stem cell proliferation. Q. Hu, B. Tanasa*, M. Trabucchi*, W. Li, J. Zhang, K.A. Ohgi, D.W. Rose, C.K. Glass, M.G. Rosenfeld (2012) Nature structural & molecular biology 19, 1168-1175 3. PI3K/AKT signaling determines a dynamic switch between distinct KSRP functions favoring skeletal myogenesis. P. Briata, W.J. Lin, M. Giovarelli, M. Pasero, C.F. Chou, M. Trabucchi, M.G. Rosenfeld, C.Y. Chen, R. Gherzi (2012) Cell Death Differ 19, 478-87 4. The RNA-binding Protein KSRP Promotes the Biogenesis of a Subset of miRNAs M. Trabucchi, P. Briata, M.F. Garcia-Mayoral, A.D. Haase, W. Filipowicz, A. Ramos, R. Gherzi, M.G. Rosenfeld (2009) Nature 459, 1010-1014. 5. LPS induces KH-type splicing regulatory protein-dependent processing of microRNA-155 precursors in macrophages T. Ruggiero*, M. Trabucchi*, F. De Santa, S. Zupo, B.D. Harfe, M.T. McManus, M.G. Rosenfeld, P. Briata, R. Gherzii (2009) FASEB J 23, 2898-2908 Awards 2011: AVENIR 2005: American-Italian Cancer Foundation award 2006: Italian Telethon foundation
-
Van Obberghen Emmanuel – Ageing and diabetes
VAN OBBERGHEN Emmanuel

VAN OBBERGHEN Emmanuel, UNS Group Leader IRCAN vanobbeg@unice.fr 0493377785 http://ircan.org 
Aging and diabetes
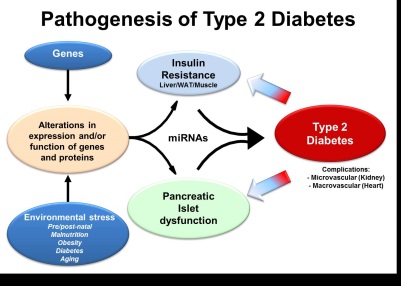 Pathogenesis of type 2 diabetes
Pathogenesis of type 2 diabetesDiabetes and cardiovascular diseases increase with age and are tightly linked as diabetes predisposes to cardiac failure, and as cardiovascular complications cause the chief diabetic morbidity. Environmental/lifestyle and genetic factors participate in the defects of type 2 diabetes (T2D), which are insufficient insulin secretion and action. The risk of chronic diseases, such as T2D, in human adults is accounted for partly by environmental influences, particularly those occurring in utero. We hypothesize that under deleterious in utero conditions the altered miRNA control on gene regulation increases the risk of insulin resistance and T2D in adults. Hence, we study offsprings of pregnant rats exposed to an inappropriate diet or environment, predisposing to insulin resistance and T2D. Offsprings are studied for miRNAs misexpressed in the liver and pancreas and for their cardiac development.As the age-related T2D increase involves pancreatic failure, we investigate whether a perturbed dialogue between beta cells and endothelial cells occurs. Finally, because both aging and diabetes favor myocardial infarction, myocardium repair is studied. Our multipronged approach will advance the understanding of the mechanisms underlying increased diabetes incidence during aging and offer key medical implications by fostering prevention strategies for T2D and its cardiovascular complications. Selected Publications
1. microRNAs and metabolism crosstalk in energy homeostasis Dumortier O, Hinault C, Van Obberghen E (2013) Cell Metabolism, in press 2. Methylglyoxal impairs insulin signalling and insulin action on glucose-induced insulin secretion in the pancreatic beta cell line INS-1E Fiory F, Lombardi A, Miele C, Giudicelli J, Beguinot F, Van Obberghen E (2011) Diabetologia 54, 2941-2952 3. Peroxisome proliferator-activated receptor β/δ (PPAR β/δ) is highly expressed in liposarcoma and promotes migration and proliferation Wagner KD, Benchetrit M, Bianchini L, Michiels JF, Wagner N. (2011) J Pathol 224, 575-88 4. The podocyte protein nephrin is required for cardiac vessel formation. Hum Mol Genet Wagner N, Morrison H, Pagnotta S, Michiels JF, Schwab Y, Tryggvason K, Schedl A, Wagner KD. (2011) Hum Mol Genet. 20, 2182-94 5. miR-375 targets 3’-phosphoinositide-dependent protein kinase-1 and regulates glucose-induced biological responses in pancreatic beta-cells. El Ouaamari A, Baroukh N, Martens GA, Lebrun P, Pipeleers D, Van Obberghen E (2008) Diabetes 57, 2708-2717 Awards 2008: Jacobeus Lecture Award, NovoNordisk Foundation, Denmark 2007: Claude Bernard Award Lecture, European Association for the Study of Diabetes 2005: D. Hermann Award, Foundation for Cardiovascular Research, Institute of France 2000: Gold Medal G.B. Morgagni Prize, Faculty of Medicine, University of Padua, Italy 1985: Oskar Minkowski Award, European Association for the Study of Diabetes
-
Van Obberghen-Schilling Ellen – Adhesion Signaling and Regulation of Cell Plasticity in the Tumor Microenvironment
VAN OBBERGHEN-SCHILLING Ellen

VAN OBBERGHEN-SCHILLING Ellen, Inserm Group Leader iBV vanobber@unice.fr 04 92 07 6430 http://ibv.unice.fr/ 
Adhesion Signaling and Regulation of Cell Plasticity in the Tumor Microenvironment
 Understanding microenvironment-driven molecular pathways involved in tumor progression.
Understanding microenvironment-driven molecular pathways involved in tumor progression.Our research is focused on adhesion-based signaling in head and neck cancer and glioblastomas. Ongoing projects address i) the molecular mechanismis underlying tumor-stroma interactions and their impact on tumor angiogenesis and invasion and ii) mechanisms that regulate the stem-cell state of cancer-initiating cells. The team has a history of productive interactions and collaborations with clinical partners. Projects rely on the integration of data derived from cellular and molecular analyses of cultured cells, orthotopic manipulations and analysis of human tumors. Selected Publications
1. Tumorigenic Potential of miR-18A* in Glioma Initiating Cells Requires NOTCH-1 Signaling. Turchi L, Debruyne DN, Almairac F, Virolle V, Fareh M, Neirijnck Y, Burel-Vandenbos F, Paquis P, Junier MP, Van Obberghen-Schilling E, Chneiweiss H, Virolle T (2013) Stem Cells, in press 2. Contrasted outcomes to gefitinib on tumoral IGF1R expression in head and neck cancer patients receiving postoperative chemoradiation (GORTEC trial 2004-02). Thariat J, Bensadoun RJ, Etienne-Grimaldi MC, Grall D, Penault-Llorca F, Dassonville O, Bertucci F, Cayre A, De Raucourt D, Geoffrois L, Finetti P, Giraud P, Racadot S, Morinière S, Sudaka A, Van Obberghen-Schilling* E, Milano G* (*equal contribution) (2012) Clin Cancer Res 18, 5123-5133 3. The miR 302-367 cluster drastically affects self-renewal and infiltration properties of glioma-initiating cells through CXCR4 repression and consequent disruption of the SHH-GLI-NANOG network. Fareh M, Turchi L, Virolle V, Debruyne D, Almairac F, de-la-Forest Divonne S, Paquis P, Preynat-Seauve O, Krause KH, Chneiweiss H, Virolle T (2012) Cell Death Differ 19, 232-244 4. Fibronectin and tenascin-C: accomplices in vascular morphogenesis during development and tumor growth. Van Obberghen-Schilling E, Tucker RP, Saupe F, Gasser I, Cseh B, Orend G (2011) Int J Dev Biol 55, 511-525 5. Autocrine fibronectin directs matrix assembly and crosstalk between cell-matrix and cell-cell adhesion in vascular endothelial cells. Cseh B, Fernandez-Sauze S, Grall D, Schaub S, Doma E, Van Obberghen-Schilling E (2010) J Cell Sci 123, 3989-3999 Awards 2001: INSERM Research Award in Physiology/Pathology 1996: Award Fondation pour la Recherche Médicale